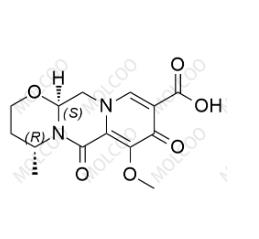
Product Number: D021034
English Name: Dolutegravir Impurity 34
English Alias: (4R,12aS)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H-pyrido[1',2':4,5]pyrazino[2,1-b][1,3]oxazine-9-carboxylic acid
CAS Number: 1335210-34-8
Molecular Formula: C₁₄H₁₆N₂O₆
Molecular Weight: 308.29
Product Advantages:
High purity and chiral confirmation:HPLC purity ≥99.0%, with chiral configuration confirmed by Chiral HPLC (optical purity ≥99.5%) and circular dichroism (CD), meeting ICH Q6A requirements for chiral impurity reference standards.
Excellent stability:Stable for 36 months when stored at -20°C in the dark, with a degradation rate <0.8% after 14 days at room temperature in solution (e.g., acetonitrile-water system), suitable for long-term quality control and stability studies of complex chiral drugs.
Clear structural characteristics:Contains a heterocyclic structure with bicyclic fusion and chiral centers, enabling precise tracking of process risks from insufficient stereoselectivity in chiral cyclization reactions or incorrect methoxylation positions in dolutegravir synthesis.
Applications:
Chiral impurity detection:Used for chiral LC-MS/MS detection of Impurity 34 in dolutegravir APIs, controlling its content ≤0.1% in accordance with ICH Q3A standards to ensure chiral impurities meet quality specifications for anti-HIV drugs.
Synthesis process optimization:In chiral cyclization reactions, monitoring impurity content (e.g., reducing impurity from 1.2% to 0.1% when changing the chiral catalyst to (S)-proline derivative) optimizes stereoselectivity and methoxylation conditions to reduce diastereomer formation.
Analytical method development:Serves as a chiral bicyclic impurity reference standard for establishing specific detection methods, such as 2D chiral HPLC (first dimension separates chiral isomers, second dimension separates structural analogs), achieving baseline separation and accurate quantification of complex chiral impurities.
Toxicological difference assessment:Provides samples for evaluating the impact of chiral configuration on toxicity, facilitating in vitro HIV integrase binding assays and in vivo embryo-fetal development toxicity studies to meet FDA requirements for in-depth stereoisomer safety assessment.
Background Description:
Dolutegravir Impurity 34 is a chiral bicyclic impurity introduced during dolutegravir synthesis due to insufficient stereocontrol in chiral cyclization reactions or poor selectivity of methoxylating reagents. Its (4R,12aS) configuration differs from the chiral centers of the parent drug, potentially affecting the drug's binding mode to HIV integrase and metabolic pathways, and increasing potential toxicity risks. According to the ICH Q6A guideline, chiral drugs require strict control of enantiomeric and diastereomeric impurities. Current industry standards set the individual impurity limit at ≤0.1%.
Research Status:
Breakthrough in detection technology:Chiral LC-MS/MS combined with a tandem cyclodextrin chiral column (e.g., Chiralpak IB) and C18 analytical column uses gradient elution to achieve simultaneous separation of chiral isomers and structural analogs, with a detection limit (LOD) as low as 0.002ppm, providing a new method for trace chiral impurity analysis.
Formation mechanism analysis:Impurity formation is closely related to the enantioselectivity of chiral catalysts (e.g., increased impurity formation with (R)-binaphthol) and the steric hindrance of methoxylating reagents. Introducing metal chiral complex catalysts (e.g., Cu(Ⅱ)-salen complex) and low-temperature (-10℃) reactions can reduce impurity formation by over 95%.
Safety evaluation progress:In vitro antiviral activity tests show that the impurity has only 3% of the HIV integrase inhibitory activity of dolutegravir, but rib fusion abnormalities were observed in high-dose groups (150mg/kg) during rat teratogenicity tests, suggesting that reasonable limits (e.g., ≤0.06%) should be set based on toxicological data to ensure drug safety.





 China
China