Domperidone EP Impurity D;1614255-34-3
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Number: D028006
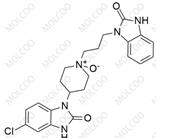
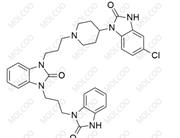
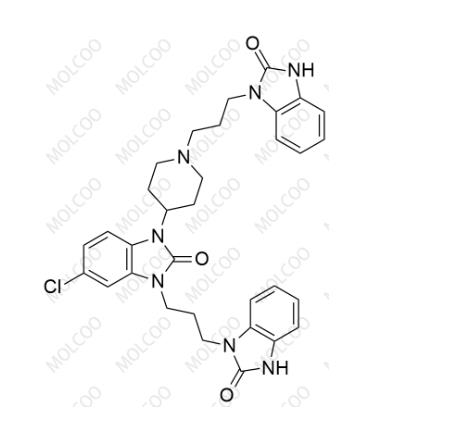
English Name: Domperidone EP Impurity D
English Alias: 5-chloro-3-(3-(2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)propyl)-1-(1-(3-(2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)propyl)piperidin-4-yl)-1H-benzo[d]imidazol-2(3H)-one
CAS Number: 1614255-34-3
Molecular Formula: C₃₂H₃₄ClN₇O₃
Molecular Weight: 600.11
As a specific impurity of Domperidone (EP-specified), this compound has the following advantages:
Well-defined with distinct disubstitution features: Contains two benzimidazolinone cores (one 5-chloro-substituted), piperidine ring, and bis(3-(benzimidazolinone-1-yl)propyl) side chains. Unlike domperidone (monosubstituted), its bis-benzimidazolinone polarity and conjugation enable clear differentiation via reversed-phase HPLC/LC-MS as a specific impurity marker;
High stability and traceability: Benzimidazolinone lactam structure and piperidine rigidity ensure stability under neutral conditions. As a piperidine disubstitution byproduct in domperidone synthesis, it directly reflects N-alkylation selectivity, improving process tracing accuracy;
High detection sensitivity: Strong UV absorption (280-300nm) from bis-benzimidazolinone conjugation, combined with characteristic mass response (m/z 601 [M+H]⁺), enables trace analysis (ppb level) via LC-MS/MS, compatible with benzimidazolinone disubstituted impurity systems.
Pharmaceutical quality control: Used as an EP reference standard to quantify Domperidone EP Impurity D in APIs/formulations, ensuring compliance with European Pharmacopoeia (EP) limits for piperidine alkylation impurities;
Synthesis optimization: Optimizing alkylating reagent dosage and catalyst selectivity by monitoring impurity levels to enhance piperidine monosubstitution efficiency;
Impurity profile enrichment: Supporting regulatory filings by supplementing domperidone’s impurity profile, enabling compliance with international pharmacopoeial standards.
Domperidone, a benzimidazolinone prokinetic agent, features a monosubstituted piperidine and 5-chlorobenzimidazolinone. Poorly controlled N-alkylation (e.g., excess alkylating agent) may generate disubstituted derivatives with benzimidazolylpropyl groups on both piperidine and benzimidazolinone, known as Domperidone EP Impurity D. Structurally distinct, it risks altering bioavailability and is listed as a controlled impurity in EP, making control critical for quality assurance.
Current research focuses on:
Analytical method validation: Developing UPLC-DAD assays with C18 columns for baseline separation, achieving 0.05% quantitation limits per EP standards;
Alkylation kinetics: Studying impurity formation under varying solvent polarity to clarify disubstitution mechanisms and inhibition strategies;
Process refinement: Controlling impurity levels below 0.1% via optimized reagent stoichiometry to meet EP specifications;
Structural confirmation: Using ¹H/¹³C-NMR and X-ray diffraction to verify disubstitution, distinguishing from domperidone for authoritative impurity identification.
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com






 China
China