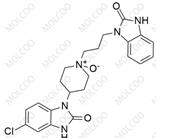
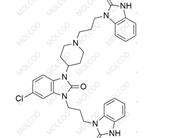
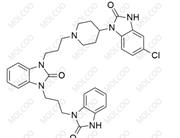
Domperidone EP Impurity D;1346599-76-5

WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Number: D028007
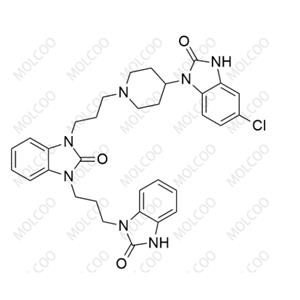
English Name: Domperidone EP Impurity E
English Alias: 1-(3-(4-(5-chloro-2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)piperidin-1-yl)propyl)-3-(3-(2-oxo-2,3-dihydro-1H-benzo[d]imidazol-1-yl)propyl)-1H-benzo[d]imidazol-2(3H)-one
CAS Number: 1346602-50-3
Molecular Formula: C₃₂H₃₄ClN₇O₃
Molecular Weight: 600.11
As a specific impurity of Domperidone (EP-specified), this compound has the following advantages:
Well-defined with distinct tricyclic features: Contains three benzimidazolinone cores (one 5-chloro-substituted), piperidine ring, and bis(3-benzimidazolylpropyl) side chains, differing from domperidone by an additional benzimidazolinone ring. The tricyclic conjugated system enables clear differentiation via reversed-phase HPLC/LC-MS as a specific impurity marker;
High stability and traceability: Tricyclic lactam structures and piperidine rigidity ensure stability under neutral conditions. As an over-coupling byproduct in domperidone synthesis, it directly reflects excessive N-alkylation, improving process tracing accuracy;
High detection sensitivity: Strong UV absorption (270-290nm) from tricyclic conjugation, combined with characteristic mass response (m/z 601 [M+H]⁺), enables trace analysis (ppb level) via LC-MS/MS, compatible with polycyclic benzimidazolinone impurity systems.
Pharmaceutical quality control: Used as an EP reference standard to quantify Domperidone EP Impurity E in APIs/formulations, ensuring compliance with EP standards for benzimidazolinone-piperidine coupling impurities;
Synthesis optimization: Optimizing reagent ratios and reaction time by monitoring impurity levels to reduce over-coupling and enhance target monocyclic coupling efficiency;
Impurity profile enrichment: Supporting regulatory filings by supplementing domperidone’s impurity profile to meet international pharmacopoeial requirements.
Domperidone, a prokinetic agent, consists of 5-chlorobenzimidazolinone, piperidine, and a single side chain. Poorly controlled coupling (e.g., excess reagents) may cause multi-step conjugation, generating tricyclic benzimidazolinone derivatives like Domperidone EP Impurity E. Pharmacologically inactive and listed as a controlled impurity in EP, its control is critical for domperidone quality assurance.
Current research focuses on:
Analytical method validation: Developing UPLC-MS/MS assays with C18 columns for baseline separation, achieving 0.05% quantitation limits per EP standards;
Coupling kinetics: Studying impurity formation under varying reaction durations to clarify over-conjugation mechanisms and inhibition strategies;
Process refinement: Controlling impurity levels below 0.1% via optimized reaction parameters to meet EP specifications;
Structural confirmation: Using 2D-NMR and HRMS to verify tricyclic structure, distinguishing from domperidone for authoritative impurity identification.
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com





 China
China