Dotinurad Impurity 68
Product Code: D081068
English Name: Dotinurad Impurity 68
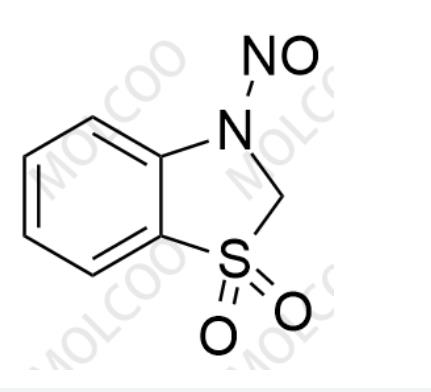
English Alias: 3-nitroso-2,3-dihydrobenzo[d]thiazole 1,1-dioxide
CAS No.:39884-80-5
Molecular Formula: C₇H₆N₂O₃S
Molecular Weight: 198.20
High-purity reference standard suitable for qualitative and quantitative analysis of drug impurities, ensuring the accuracy and reliability of test results.
Well-defined structure with traceable synthesis routes, meeting the strict requirements of drug R&D, quality control, and regulatory submissions.
Primarily used for impurity analysis of the gout treatment drug Dotinurad, serving as a reference standard in detection methods such as HPLC and LC-MS to monitor impurity residues in the drug production process.
Supports pharmaceutical quality research to help pharmaceutical companies comply with the impurity limit requirements of domestic and international regulatory agencies (e.g., FDA, EMA, NMPA).
Dotinurad is a selective uric acid reabsorption inhibitor that promotes uric acid excretion by inhibiting the uric acid transporter URAT1, used for the treatment of hyperuricemia and gout. During drug synthesis and storage, various impurities may be generated, among which Impurity 68 is a potential genotoxic or process-related impurity that requires strict control of its content. The study of this impurity is a key part of drug quality evaluation, directly related to the safety and efficacy of the drug.
Current research on Dotinurad Impurity 68 mainly focuses on the following aspects:
Development of analytical methods: Establishing specific and sensitive detection methods (such as UPLC-MS/MS) to achieve precise determination of trace impurities.
Toxicological evaluation: Assessing the potential toxicity of the impurity through in vitro and in vivo tests to provide data support for setting safety thresholds.
Process optimization: Studying the formation mechanism of impurities and reducing their generation through improved synthesis routes or purification processes to ensure the controllability of the drug production process.
Compliance with international standards: Referencing international guidelines such as ICH Q3A/B/C to classify impurities and set limits, promoting the international registration process of drugs.