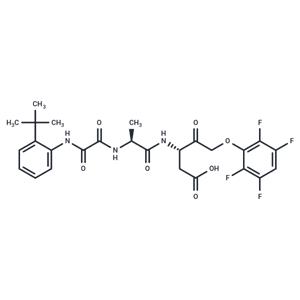
| Name | Emricasan |
| Description | Emricasan (IDN-6556) is a pan-caspase inhibitor with irreversible properties. Emricasan has anti-inflammatory and anti-apoptotic activity and can be used in the treatment of infections and liver failure, etc. Emricasan also inhibits Zika virus infections. |
| Cell Research | Astrocytes are mock-infected, treated with DMSO or treated with 2 μM niclosamide, 92 μM PHA-690509, 9 μM emricasan, or a combination of 92 μM PHA-690509 and 9 μM emricasan for 1 h before infection with PRVABC59 (MOI = 0.5). Cells are fixed 24 h after infection and stained for ZIKVE and nuclei.(Only for Reference) |
| In vitro | METHODS: Primary rat cirrhotic hepatocytes were treated with Emricasan (50 µM) for 24 h, and mRNA expression levels were measured.
RESULTS: Emricasan directly improved the expression of hepatocyte-specific markers. [1]
METHODS: Human cervical cancer cells HeLa were pretreated with Emricasan (10 µM) for 1 h, then treated with vincristine (20 nM) for 44 h. Cell death was detected by Flow Cytometry.
RESULTS: Emricasan eliminated 95% of vincristine-mediated cell death. [2] |
| In vivo | METHODS: To study the effects on chronic liver disease, Emricasan (10 mg/kg, 0.9% dimethylcarboxycellulose) was administered orally once daily for seven days to rats with advanced cirrhosis due to chronic CCl4 administration.
RESULTS: Emricasan ameliorated hepatic sinusoidal microvascular dysfunction in cirrhotic patients, resulting in significant improvement in fibrosis, portal hypertension and liver function. [1]
METHODS: To investigate the effects on cirrhosis, Emricasan (10 mg/kg) was administered intraperitoneally once daily for 10-20 days to C57BL/6 mice with secondary biliary cirrhosis induced by bile duct ligation (BDL).
RESULTS: Emricasan treatment improved survival and portal hypertension (PHT) in a mouse model of long-term BDL. [3] |
| Storage | Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | H2O : < 1 mg/mL (insoluble or slightly soluble)
DMSO : 257.5 mg/mL (452.15 mM), Sonication is recommended.
Ethanol : 93 mg/mL (163.3 mM), Sonication is recommended.
10% DMSO+40% PEG300+5% Tween 80+45% Saline : 5 mg/mL (8.78 mM), Solution.
|
| Keywords | PF-03491390 | PF03491390 | Inhibitor | inhibit | InfluenzaVirus | Influenza Virus | IDN6556 | IDN 6556 | Emricasan | Caspase |
| Inhibitors Related | Acetylcysteine | Nitazoxanide | Penicillin G potassium | Curcumin | Magnesium acetate tetrahydrate | (E)-3-(p-Tolyl)acrylaldehyde | Cystamine dihydrochloride | Copper Sulfate Pentahydrate | Baicalein | Salcomine | Benzimidazole | β-Cyclodextrin |
| Related Compound Libraries | Highly Selective Inhibitor Library | Failed Clinical Trials Compound Library | Bioactive Compound Library | Membrane Protein-targeted Compound Library | Anti-Cancer Clinical Compound Library | Drug Repurposing Compound Library | Neuroprotective Compound Library | Inhibitor Library | Immunology/Inflammation Compound Library | Anti-Aging Compound Library | Anti-Cancer Active Compound Library | Anti-Cancer Drug Library |

 United States
United States