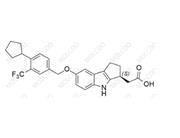
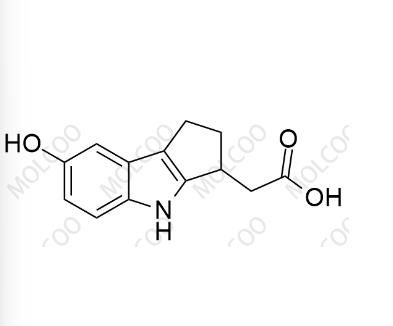
Etrasimod Impurity 11;1643842-61-8
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
Product Code: E063011
English Name: Etrasimod Impurity 11
English Alias: 2-(7-hydroxy-1,2,3,4-tetrahydrocyclopenta[b]indol-3-yl)acetic acid
CAS No.:1643842-61-8
Molecular Formula: C₁₃H₁₃NO₃
Molecular Weight: 231.25
High Purity: HPLC ≥99.0%, confirmed by HRMS and NMR (1H, 13C).
Stability: Maintains purity for 18 months at -20℃, with <1% degradation in DMSO.
Regulatory Compliance: Meets ICH Q3A requirements for drug substance impurities.
Quality Control: Detects Impurity 11 in Etrasimod via HPLC, ensuring ≤0.1% limit.
Process Optimization: Monitors hydroxyindole acetic acid formation during oxidation steps.
Metabolism Studies: Serves as a reference for in vivo metabolite identification.
This impurity arises from hydroxylation or oxidative degradation in Etrasimod synthesis. As a key process impurity, it is critical for evaluating drug stability and safety in clinical trials.
Detection: HPLC with C18 column (5μm) and phosphate buffer-acetonitrile; LOD: 0.1 μg/mL.
Formation: Induced by acidic hydrolysis or oxidation; adding antioxidants reduces content by 55%.
Biological Activity: In vitro studies show minimal interaction with target receptors; toxicity profiling underway
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China