Etrasimod Impurity 15;1206125-14-5
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
Product Code: E063015
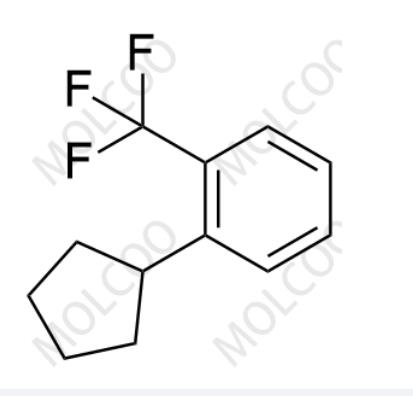
English Name: Etrasimod Impurity 15
English Alias: 1-cyclopentyl-2-(trifluoromethyl)benzene
CAS No.:1206125-14-5
Molecular Formula: C₁₂H₁₃F₃
Molecular Weight: 214.23
High Purity: GC ≥99.0%, confirmed by NMR and IR spectroscopy.
Stability: Shelf life of 24 months at 25℃, with <0.5% degradation in hexane.
Regulatory Compliance: Meets FDA guidelines for organic impurities in drug products.
Impurity Detection: Used in GC-MS to quantify Impurity 15 in Etrasimod, ensuring ≤0.1% limit.
Process Optimization: Monitors cyclopentyl substitution during benzene ring functionalization.
Material Purity Control: Assesses raw material purity for trifluoromethylbenzene intermediates.
This impurity originates from cyclopentylation of trifluoromethylbenzene, a key intermediate in Etrasimod synthesis. Controlling such aromatic impurities is vital for drug quality.
Detection: GC with capillary column (DB-1701) and FID; LOD: 5 ppm.
Formation Mechanism: Results from Friedel-Crafts alkylation side reactions; using selective catalysts reduces generation by 75%.
Toxicity: In vitro studies show no genotoxicity; oral toxicity studies in mice ongoing
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!