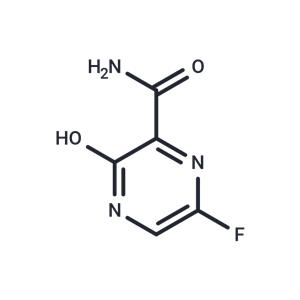
| Name | Favipiravir |
| Description | Favipiravir is a potent and selective RNA-dependent RNA polymerase inhibitor for the treatment of influenza virus infections. |
| Cell Research | The cytotoxicity of T-705 is evaluated by an assay with XTT. XTT is converted to aqueous formazan by an enzyme in MDCK cells, Vero cells, HEL cells, A549 cells, HeLa cells, and HEp-2 cells. The compounds are diluted to the appropriate concentrations (volume, 100 μl) with test medium (EMEM containing 10% FCS) in 96-well culture plates in which each well contains a concentration of 2 × 103 cells/100 μL. The test plates are incubated for 3 days at 37°C in 100% humidity and 5% CO2. After 3 days, 50 μl of the XTT reagent (1 mg/ml in FCS-free EMEM containing 5 mM phenazine methosulfate) is added, and the reaction product is assayed by measurement of the absorbance at 450 nm with a microplate reader. Cytotoxicity is expressed as the 50% cell-inhibitory concentration (CC50).(Only for Reference) |
| In vitro | METHODS: RAW 264.7 cells were infected with MNV at an MOI of 0,001 using a T-705 dilution series (3.13-200 lg/mL). The MTS CPE reduction method was used to determine the antiviral activity of T-705 in the MNV/RAW 264.7 cell line. After 3 days of incubation, complete CPE was observed in the infected untreated cells. The cell culture supernatant was collected and analyzed by quantitative RT- PCR (qRT-PCR) quantitatively detects viral RNA load.
RESULTS T-705 inhibited MNV-induced CPE (EC50: 39 ± 4 lg/mL [250 ± 11 lM]) and MNV RNA synthesis in cell culture (EC50: 19 ± 6 lg/mL [124 ± 42 lM]). [2] |
| In vivo | METHODS: When Favipiravir (T-705) is used at a dose of 1-100mg/kg and infected with a lethal dose of influenza virus A/Victoria/3/75 (H3N2), A/Osaka/5/70 (H3N2) or A/ Duck/MN/1525/81 (H5N1) was administered orally 2 or 4 times a day to mice for 5 days, and the survival rate of the mice was observed.
RESULTS Favipiravir (T-705) showed improved survival compared with placebo at doses of 30 mg/kg/day or higher, the drug was also administered at doses of 33 mg/kg/day or higher Provides significant protection against A/Duck/MN/1525/81 (H5N1) virus. [1]
METHODS: Favipiravir was administered at 120 mg/kg/day or 200 mg/kg/day on days 1-5 after lethal SFTSV infection in type I interferon receptor knockout (IFNAR −/−) mice. (T-705) and continued for 5 days to observe the growth status of the mice.
RESULTS All favipiravir-treated mice at doses of 120 mg/kg/day or 200 mg/kg/day, respectively, survived lethal SFTSV infection when treatment was initiated within 3 and 4 days of infection. [3] |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year
Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | 10% DMSO+40% PEG300+5% Tween 80+45% Saline : 4 mg/mL (25.46 mM), Sonication is recommended.
Ethanol : 12 mg/mL (76.38 mM), Sonication is recommended.
H2O : 5 mg/mL (31.83 mM), Sonication is recommended.
DMSO : 126 mg/mL (802.04 mM), Sonication is recommended.
|
| Keywords | T705 | T 705 | SARS-CoV | SARSCoV | SARS coronavirus | RNASynthesis | RNA-dependent RNA polymerase | RNA Synthesis | Inhibitor | inhibit | InfluenzaVirus | Influenza Virus | Favipiravir | DNASynthesis | DNA/RNA Synthesis | DNA Synthesis |
| Inhibitors Related | Neomycin sulfate | Levulinic acid | D(+)-Raffinose pentahydrate | Guanidine hydrochloride | Sulfamethoxazole sodium | Terbinafine hydrochloride | Hydroxychloroquine | Thymidine | Hyaluronic acid sodium (MW 20 kDa) | Dimethyl sulfoxide | Sodium diacetate | Sodium bicarbonate |
| Related Compound Libraries | Bioactive Compound Library | ReFRAME Related Library | Anti-Cancer Clinical Compound Library | Anti-Viral Compound Library | Drug Repurposing Compound Library | Inhibitor Library | Anti-Cancer Approved Drug Library | Immunology/Inflammation Compound Library | Anti-Aging Compound Library | Bioactive Compounds Library Max | Anti-Cancer Compound Library | Anti-Cancer Drug Library |

 United States
United States