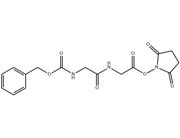
Peptide Synthesis:Fmoc-Gly-OSu acts as a protected amino acid, allowing for the sequential addition of other amino acid residues during peptide synthesis. By removing the Fmoc protecting group and attaching the next amino acid, peptides with specific sequences can be efficiently synthesized.
Protein Labeling and Conjugation:The succinimide ester moiety of Fmoc-Gly-OSu reacts with the amino groups of proteins, enabling their labeling and conjugation. This conjugation reaction is useful for preparing antibody-peptide conjugates, protein-peptide hybrids, and other bioconjugates for biological analysis, drug screening, and protein function studies.
Bioconjugation and Surface Modification:Fmoc-Gly-OSu can be used as a linker to attach biological molecules (such as proteins, nucleic acids) to solid supports, nanoparticles, or biosensors. Through its reaction with the amino groups of the biomolecule, directed immobilization and surface modification can be achieved. | 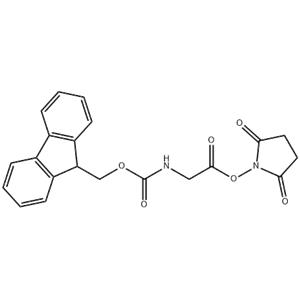

 China
China