Features
Designed under ISO 9001:2015 and ISO 13485:2016
Manufactured and QC tested under a GMP compliance factory
FDA DMF filed
Animal-Free materials
Beta-lactam materials free
Batch-to-batch consistency
Stringent quality control tests
Source
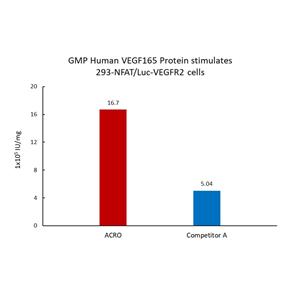
GMP Human VEGF165 Protein (GMP-VE5H23) is expressed from human 293 cells (HEK293). It contains AA Ala 27 - Arg 191.
Predicted N-terminus: Ala 27
Molecular Characterization

Endotoxin
Less than 10 EU/mg, tested by the LAL method in compliance with USP <85> and Ph. Eur. 2.6.14.
Host Cell Protein
<0.5 ng/µg of protein tested by ELISA.
Host Cell DNA
<0.02 ng/μg of protein tested by qPCR.
Sterility
Sterility testing was performed using the membrane filtration method in compliance with USP <71> and Ph. Eur. 2.6.1.
Mycoplasma
Negative
Purity
>95% as determined by SDS-PAGE.
Formulation
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with protectants.
Contact us for customized product form or formulation.
Vial Specification
2R (13 mm neck finish)
Shipping
This product is supplied and shipped with blue ice, please inquire the shipping cost.
Storage
Upon receipt, store it immediately at -20°C or lower for long term storage.
Please avoid repeated freeze-thaw cycles.
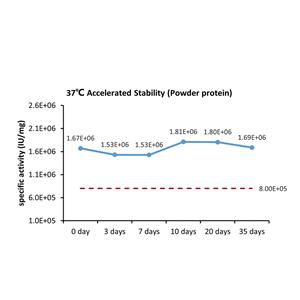
This product is stable after storage at:
-20°C to -70°C for 5 years in lyophilized state;
-70°C for 12 months under sterile conditions after reconstitution.

 China
China