| Name | H3B-8800 |
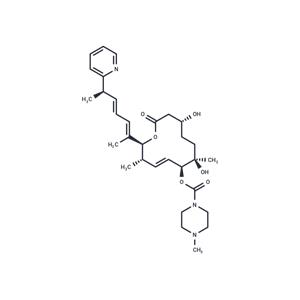
| Description | H3B-8800 is an SF3B1 modulator that can be used to study transfusion-dependent anemia. |
| In vivo | We conducted a Phase I clinical trial of H3B-8800, an oral small molecule compound that binds splicing factor 3B1 (SF3B1), in patients with MDS, CMML or AML. Of the 84 patients enrolled in the trial (42 MDS, 4 CMML and 38 AML), 62 were dependent on red blood cell (RBC) transfusions at enrollment. The dose-escalation cohort examined two once-daily dosing regimens: regimen I (5 days on/9 days off, study dose range 1-40 mg, n=65) and regimen II (21 days on/7 days off, 7-20 mg, n=19); 27 patients were treated for ≥180 days. The most common treatment-related emergent adverse events included diarrhea, nausea, fatigue, and vomiting. No complete or partial responses meeting IWG criteria were observed; however, RBC transfusion free intervals of more than 56 days were observed in nine patients who were transfusion dependent at enrollment (15%). Five of 15 MDS patients with missense SF3B1 mutations experienced RBC transfusion independence (TI). Increased expression of the pre-treatment target of splicing transmembrane protein 14C (TMEM14C) was observed in MDS patients experiencing RBC TI. |
| Storage | Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | DMSO : 122.5 mg/mL (220.44 mM), Sonication is recommended.
|
| Keywords | SF3B1 | H3B-8800 | H3B8800 | H3B 8800 |
| Inhibitors Related | Stavudine | Aceglutamide | Urea | Tamoxifen | Cysteamine hydrochloride | Metronidazole | Citric Acid Triammonium | Formamide | Dimethyl phthalate | Alginic acid | Sodium Molybdate | Sildenafil citrate |
| Related Compound Libraries | Failed Clinical Trials Compound Library | Bioactive Compound Library | Hematonosis Compound Library | Anti-Cancer Clinical Compound Library | Drug Repurposing Compound Library | Clinical Compound Library | Bioactive Compounds Library Max | Anti-Cancer Compound Library | Anti-Cancer Active Compound Library | Anti-Cancer Drug Library |

 United States
United States