Synonym
CLD1, ILVASC, SEMP1
Source
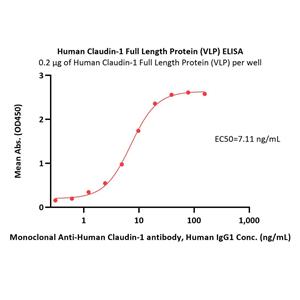
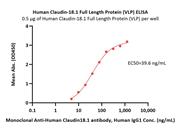
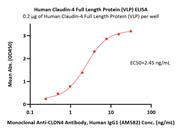
Human Claudin-1 Full Length Protein (VLP) (CL1-H52P6) is expressed from human 293 cells (HEK293). It contains AA Met 1 - Val 211 (Accession # O95832).
Predicted N-terminus: Met 1
Molecular Characterization
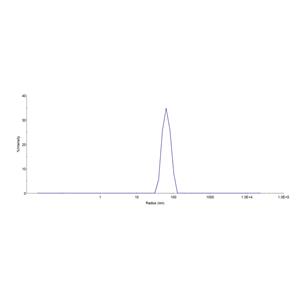
Virus-like particles(VLPs) are formed by self-assembly of capsid proteins from viruses. Membrane Proteins can be constituted in-situ with VLPs produced from HEK293 cell cultures. These VLPs concentrate conformationally intact membrane proteins directly on the cell surface and produce soluble, high-concentration proteins perfect for immunization and antibody screening.

The VLPs provide the display of properly folded membrane proteins in their native cellular membrane in a compact size of 100~300 nm diameter (similar to the size of most viruses) making it optimal targets for dendritic cells in vivo and surface attachment for phage display.
Endotoxin
Less than 1.0 EU per μg by the LAL method / rFC method.
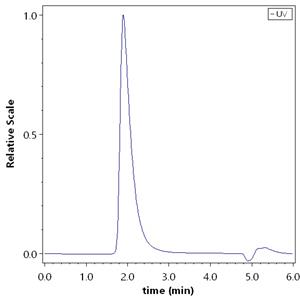
Purity
>95% as determined by SEC-HPLC.
Formulation
The VLPs are highly immunogenic, so the immunization strategy should be optimized (antigen dose, regimen and adjuvant).
Supplied as 0.2 μm filtered solution in PBS, Arginine, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
Shipping
This product is supplied and shipped with dry ice, please inquire the shipping cost.
Storage
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
The product MUST be stored at -70°C or lower upon receipt;
-70°C for 12 months under sterile conditions.
Background
Tight junctions represent one mode of cell-to-cell adhesion in epithelial or endothelial cell sheets, forming continuous seals around cells and serving as a physical barrier to prevent solutes and water from passing freely through the paracellular space. These junctions are comprised of sets of continuous networking strands in the outwardly facing cytoplasmic leaflet, with complementary grooves in the inwardly facing extracytoplasmic leaflet. The protein encoded by this gene, a member of the claudin family, is an integral membrane protein and a component of tight junction strands. Loss of function mutations result in neonatal ichthyosis-sclerosing cholangitis syndrome. [provided by RefSeq, Jul 2008]

 China
China