Synonym
CLDN9
Source
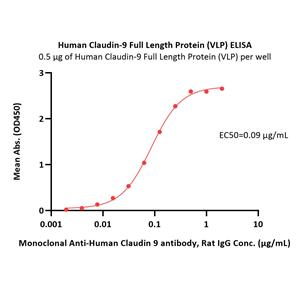
Human Claudin-9 Full Length Protein-VLP (CL9-H52P5) is expressed from human 293 cells (HEK293). It contains AA Met 1 - Val 217 (Accession # O95484-1).
Predicted N-terminus: Met 1
Molecular Characterization
Virus-like particles(VLPs) are formed by self-assembly of capsid proteins from viruses. Membrane Proteins can be constituted in-situ with VLPs produced from HEK293 cell cultures. These VLPs concentrate conformationally intact membrane proteins directly on the cell surface and produce soluble, high-concentration proteins perfect for immunization and antibody screening.

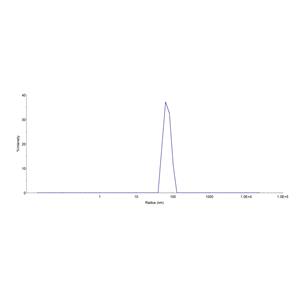
The VLPs provide the display of properly folded membrane proteins in their native cellular membrane in a compact size of 100~300 nm diameter (similar to the size of most viruses) making it optimal targets for dendritic cells in vivo and surface attachment for phage display.
Endotoxin
Less than 1.0 EU per μg by the LAL method / rFC method.
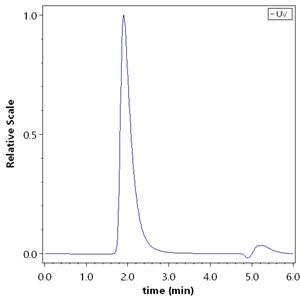
Purity
>95% as determined by SEC-HPLC.
Formulation
The VLPs are highly immunogenic, so the immunization strategy should be optimized (antigen dose, regimen and adjuvant).
Supplied as 0.2 μm filtered solution in PBS, Arginine, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
Shipping
This product is supplied and shipped with dry ice, please inquire the shipping cost.
Storage
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
The product MUST be stored at -70°C or lower upon receipt;
-70°C for 3 months under sterile conditions.
Background
Claudin-9 belongs to the claudin family. Claudins constitute integral membrane proteins responsible for solute and electrolyte permeability of the tight junction that serve as a physical barrier to prevent solutes and water from passing freely through the paracellular space between epithelial or endothelial cell sheets. Tight junctions also play a critical role in maintaining cell polarity and signal transductions. Claudin-9 creates charge specific channels in the paracellular space, plays a major role in tight junction-specific obliteration of the intercellular space, through calcium-independent cell-adhesion activity, is required to preserve sensory cells in the hearing organ because claudin-9-defective tight junctions fail to shield the basolateral side of hair cells from the K+-rich endolymph. Its ion barrier function is essential in the cochlea, but appears to be dispensable in other organs. Is one of the entry cofactors for hepatitis C virus; it enables HCV entry into target cells just as efficiently as CLDN1.

 China
China