Features and Advantages
Seamless Transition from Preclinical to Clinical: Same cell clone, process, and QC as GMP products.
Enhanced Production Standards: AOF raw materials, pharma-grade excipients, Grade B+A (ISO 5) environment.
Comprehensive Quality Control: Additional strict testing for process impurities, sterility, and mycoplasma.
Cost-Effective Solution: GMP-comparable quality at an economical price for early development.
Risk Mitigation: Enhanced safety through rigorous impurity control and comprehensive testing.
Synonym
RP1-261G23.1, MGC70609, MVCD1, VEGFA, VPF
Source
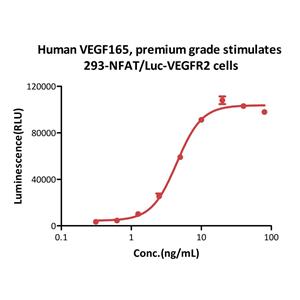
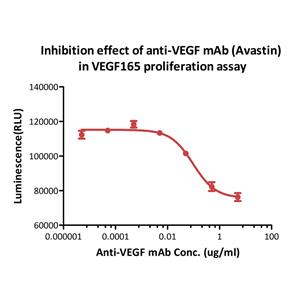
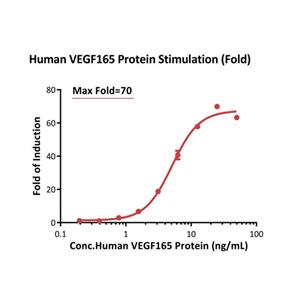
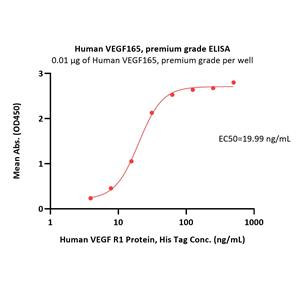
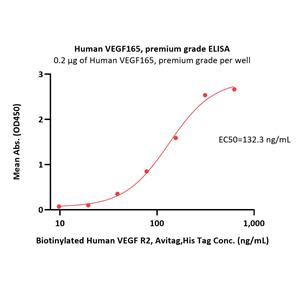
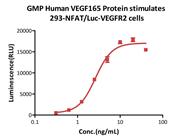
Human VEGF165, premium grade (VE5-H4210) is expressed from human 293 cells (HEK293). It contains AA Ala 27 - Arg 191.
Predicted N-terminus: Ala 27
It is produced under our rigorous quality control system that incorporates a comprehensive set of tests including sterility and endotoxin tests. Product performance is carefully validated and tested for compatibility for cell culture use or any other applications in the early preclinical stage.
GMP-VE5H23 is the GMP version of this VE5-H4210. These two proteins display indistinguishable performance profiles, thereby ensuring a seamless transition for end users from early preclinical stage to later clinical phases.
Molecular Characterization

Endotoxin
Less than 0.01 EU per μg by the LAL method / rFC method.
Host Cell Protein
<0.5 ng/µg of protein tested by ELISA.
Host Cell DNA
<0.02 ng/μg of protein tested by qPCR.
Sterility
Negative
Mycoplasma
Negative
Purity
>95% as determined by SDS-PAGE.
>95% as determined by SEC-MALS.
Formulation
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
Reconstitution
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
Storage
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
-20°C to -70°C for 24 months in lyophilized state;
-70°C for 3 months under sterile conditions after reconstitution.

 China
China