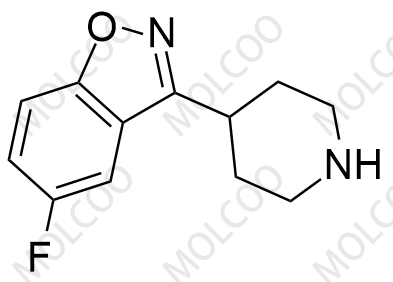
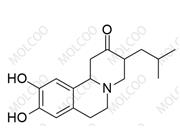
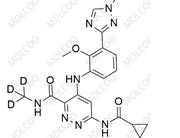
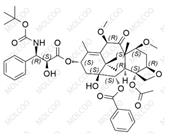
Iloperidone Impurity 84163-64-4
Product Name: Iloperidone Impurity Reference Standard
CAS No.: Including 133454-47-4 (Iloperidone API), 129938-20-1 (Impurity A), 531524-17-1 (Impurity 13), etc.
Molecular Formula & MW: Varies by impurity type (e.g., C12H13F2NO for HC102-201801, MW 225.23; C24H27FN2O4 for Impurity A, MW 426.4806).
Storage Conditions: 2-8°C (some impurities require -20°C storage).
Purity: ≥95% (some ≥98%).
Product Category: Pharmaceutical Impurity Reference Standard.
Applications: Quality control, stability studies, method development, and regulatory filing for Iloperidone API and formulations.
Key Features
Multiple Specifications: Available in 10mg, 25mg, 50mg, and 100mg packages.
Structural Confirmation: Includes COA, HPLC, NMR, and MS spectra for impurity identification.
Regulatory Compliance: Meets ICH, FDA, and NMPA requirements for drug development.
Custom Synthesis: Tailored impurity production for specific research needs.
Applications
Method Development: Validates HPLC and LC-MS methods for impurity separation.
Stability Studies: Evaluates degradation products under various conditions.
Impurity Profiling: Identifies impurity sources to optimize manufacturing processes.
Regulatory Filings: Supports pharmacopeial method validation and impurity limit setting.




 China
China