1/1
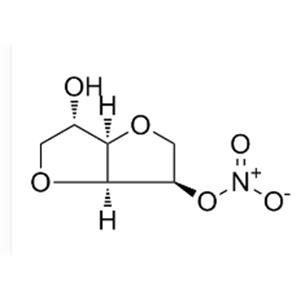
Isosorbide 5-mononitrate NEW
- Min. Order1kg
- Purity99%
- Cas No16051-77-7
- Supply Ability100kg
- Update time2025-10-13
| Product Name | Isosorbide 5-mononitrate |
| CAS No | 16051-77-7 |
| EC-No | |
| Min. Order | 1kg |
| Purity | 99% |
| Supply Ability | 100kg |
| Release date | 2025/10/13 |
PRODUCT INFORMATION
Names
| Name | isosorbide mononitrate |
|---|---|
| Synonym | More Synonyms |
Isosorbide 5-mononitrate Biological Activity
| Description | Isosorbide mononitrate(Isosorbide-5-mononitrate) is a nitrate-class compound used for angina pectoris; acts by dilating the blood vessels so as to reduce the blood pressure. |
|---|---|
| Related Catalog | Signaling Pathways >> Autophagy >> Autophagy Research Areas >> Cardiovascular Disease |
Chemical & Physical Properties
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 364.5±42.0 °C at 760 mmHg |
| Melting Point | 88-93 °C |
| Molecular Formula | C6H9NO6 |
| Molecular Weight | 191.139 |
| Flash Point | 174.2±27.9 °C |
| Exact Mass | 191.042984 |
| PSA | 93.74000 |
| LogP | -0.51 |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.543 |
| Water Solubility | soluble |
MSDS
Isosorbide 5-mononitrate MSDS(Chinese) |
Toxicological Information
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
Safety Information
| Hazard Codes | F:Flammable |
|---|---|
| Risk Phrases | R11;R36/37/38 |
| Safety Phrases | S37/39-S16-S15 |
| RIDADR | 3251 |
| RTECS | LZ4386500 |
| Packaging Group | III |
| Hazard Class | 1.1A |
| HS Code | 2932999099 |
Synthetic Route
Previous 1/4 Next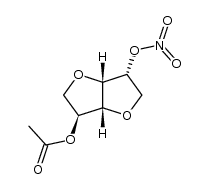 ISMN acetate 39813-48-4 ~92%  Isosorbide 5-mo... 16051-77-7 |
| Literature: Boehringer Mannheim GmbH Patent: US5614643 A1, 1997 ; |
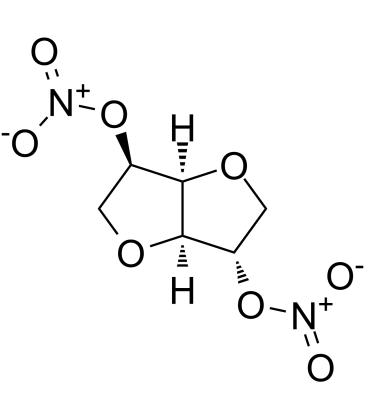 isosorbide dini... 87-33-2 ~69%  Isosorbide 5-mo... 16051-77-7 |
| Literature: Bhar, Debjani; Chandrasekaran, Srinivasan Indian Journal of Chemistry, Section B: Organic Chemistry Including Medicinal Chemistry, 1997 , vol. 36, # 9 p. 793 - 795 |
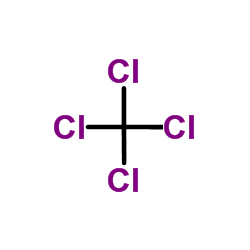 Carbon tetrachl... 56-23-5 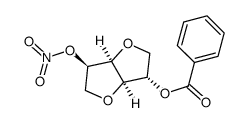 isosorbide-2-be... 75648-24-7 ~%  Isosorbide 5-mo... 16051-77-7 |
| Literature: US4297286 A1, ; |
Precursor & DownStream
| Precursor 10 | Previous 1/3 Next |
|---|---|
| |
| DownStream 0 | |
Customs
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
Company Profile
Our company is a high-tech enterprise specializing in chemical raw materials such as electronic materials, optoelectronic materials, semiconductor materials, UV monomers, silane catalysts, cosmetic raw materials, etc. The company integrates research and development, production, customized synthesis, and sales, and is committed to providing customers with high-quality chemical product solutions.
Our Company


Our Advantage
Rich experience
Our products have been exported to many countries, including Germany, Spain, the United Kingdom, the United States, Australia, the Middle East, Asia, and more. We have received highly positive feedback from our clients and have established long-term friendly cooperative relationships with them.
Excellent quality, purity, and competitive price
Excellent quality is one of the cornerstones of our success. We welcome ordering samples for quality testing.

Safe and fast delivery
We will arrange shipment of spot products as soon as we receive payment.
Customized products are determined based on the synthesis time of different products.
Excellent pre-sales and after-sales service
Pre sales:
We are committed to providing the most favorable quotes and detailed information about our products and company.
after-sale service:
We assist buyers in customs clearance by providing necessary documents and information.
If there is any dispute over product quality, we are committed to providing the best solution.
• Packaging:
1kg/bag, 25kg/drum, 50kg/drum, 180kg/drum, 200kg/drum, 1T/bag/drum, standard export packaging or packaging required by customers


• Shipping

• Delivery:
Within 7 days after receiving your payment
Contact information
For more details, pls contact us freely.
Contact name: Tina
Email address: Tina@fdachem.com
Mob: 86 13213167925
WhatsApp/Skype/Wechat/LINE: 86 15225627621
Company Profile Introduction
Our company is a high-tech enterprise specializing in chemical raw materials such as electronic materials, optoelectronic materials, semiconductor materials, UV monomers, silane catalysts, cosmetic raw materials, etc. The company integrates research and development, production, customized synthesis, and sales, and is committed to providing customers with high-quality chemical product solutions. With our agile custom synthesis capabilities and responsive supply chain system, our products have gained widespread adoption across Asian markets including Japan, South Korea, Singapore, and Malaysia, while also expanding deeply into Europe and the Americas. We have forged long-term strategic partnerships with numerous leading global enterprises in the sector. "Driving value through technology and earning trust through service" is our core mission. Looking ahead, we will continue to focus on technological breakthroughs and green innovation in new materials, collaborating with partners to build a high-quality industrial ecosystem and provide globally competitive solutions with greater foresight.

 China
China