Human Laminin 511 Protein, premium grade
The schematic diagram of Laminin in ES/iPS cell culture
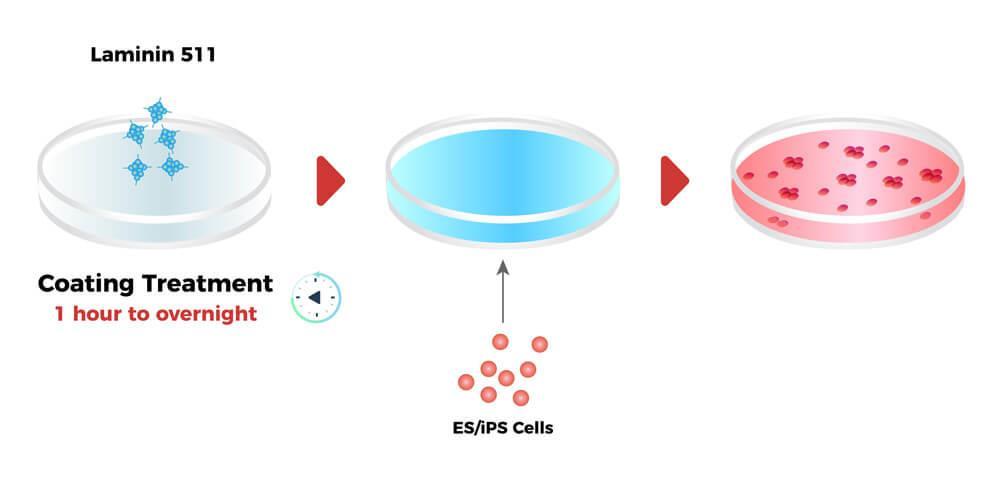
Product Details
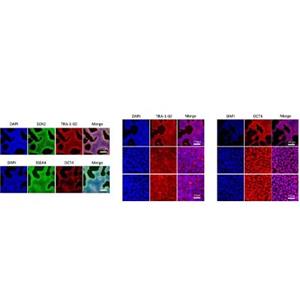
Laminin 511 E8 (LN511-E8) is a recombinant human protein that provides a defined surface for the in vitro feeder-free culture of multiple human pluripotent stem cells (PSCs). As a truncated form of laminin 511, LN511-E8 is a functionally minimal form that retains the full capability for binding to integrins. LN511-E8 has been proven to maintain normal growth characteristics and stemness in multiple PSC lines without simultaneous differentiation, which includes ESC, iPSC, MSC, etc. In addition, LN511-E8 has been demonstrated to support PSC growth for >10 passages without any signs of karyotypic abnormalities and to maintain the ability of PSCs to differentiate into all three germ line lineages. As published by Takamichi Miyazaki et al., the LN511-E8 variant of laminin 511 shows higher efficiency for supporting the adhesion of dissociated cells than did wild-type laminin 511 which makes it a cost-effective choice.
High-quality products and regulatory support files are essential for the smooth transition from preclinical research & development to cell therapy clinical study. Designed for clinical research, GMP Laminin 511 is manufactured by an animal-free process in a GMP-compliant facility. A full battery of QC testing is implemented to ensure product quality, including purity, bioactivity, sterility, mycoplasma, endotoxin, etc. GMP Laminin 511 protein (GMP-LA1H25) is the GMP version of laminin 511 protein premium grade (LA8-H5283L), and they have the same performance for a seamless transition.
This product is designed and developed utilizing our proprietary AI technology, which is protected by patent rights. We retain full commercial rights to this product.
Optimized and defined surface
Laminin 511 E8 (LN511-E8) has been proven to allow cell seeding at low density. In a feeder-free culture system, the seeded cells demonstrate high motility with higher clonal survival.
Physiology-related
The laminin 511 isoform is crucial to the growth and maintenance of hPSCs in human through its binding to cell receptors a6B1 integrin. LN511-E8 is truncated laminin 511 isoform which works in multiple stem cell lines, from iPSC to hESC and MSC.
Better Adhesion
LN511-E8 variant of laminin 511 shows higher efficiency for supporting the adhesion of dissociated cells than did wild-type laminin 511.
Enhance cell differentiation
Due to the diverse biorelevant environment, GMP Laminin 511 could also enhance cell differentiation, polarization and organization of target cell types.
Reduce Variability
LN511-E8 is a defined, recombinant human protein with better lot-to-lot consistency that reduces variability in your PSC cultures.
Features and Advantages
1. Seamless Transition from Preclinical to Clinical: Same cell clone, process, and QC as GMP products.
2. Enhanced Production Standards: AOF raw materials, pharma-grade excipients, Grade B+A (ISO 5) environment.
3. Comprehensive Quality Control: Additional strict testing for process impurities, sterility, and mycoplasma.
4. Cost-Effective Solution: GMP-comparable quality at an economical price for early development.
5. Risk Mitigation: Enhanced safety through rigorous impurity control and comprehensive testing.
Key parameter
Purity (SDS PAGE) | > 95% |
Mycoplasma Test | Negative |
Sterility Test | Negative |
Integrin Binding Assay | 1.0 nM < KD < 15 nM |
Endotoxin Test | <0.01 EU per μg |
Host Cell Protein | <0.5 ng/µg |
Host Cell DNA | <0.02 ng/μg |
Formulation
Supplied as 0.2 μm filtered solution in PBS, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
Shipping
This product is supplied and shipped with dry ice, please inquire the shipping cost.
Storage
For long term storage, the product should be stored at liquid state at -20℃ or below.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
2-8°C for 6 months under sterile conditions;
-20°C or below for 3 years.
Quality Description
ACRO's Research-grade products are suitable for a wide range of cell culture applications, particularly for research use in academic institutions. These products are sterilized by filtration before lyophilization. Typical specifications include endotoxin levels of <0.01 EU/μg and purities >95%. Biological activity is calibrated against WHO/NIBSC standards when available.
ACRO's Premium-grade (Pre-GMP) products are characterized by their high quality and enhanced safety profiles, making them ideal for early-stage discovery and manufacturing processes in cell therapy companies. A key advantage is their seamless transition to corresponding GMP-grade versions. Biological activity is calibrated against WHO/NIBSC standards when available. Typical specifications include endotoxin levels of <0.01 EU/μg and purities >95%. In addition, rigorous testing is conducted to ensure the absence of mycoplasma, HCD, and HCP, thereby guaranteeing product safety.

 China
China