Lapatinib Impurity 12;697299-82-4

WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Number: L079012
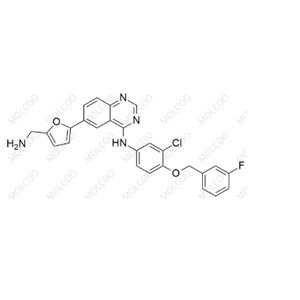
English Name: Lapatinib Impurity 12
English Alias: 6-(5-(aminomethyl)furan-2-yl)-N-(3-chloro-4-((3-fluorobenzyl)oxy)phenyl)quinazolin-4-amine
CAS Number: 697299-82-4
Molecular Formula: C26H20ClFN4O2
Molecular Weight: 474.91
Product Advantages: Lapatinib Impurity 12 has extremely high purity, and its structure is accurately confirmed by high-precision spectroscopic analysis (such as nuclear magnetic resonance and mass spectrometry). It has stable chemical properties and maintains good uniformity under different experimental conditions and storage environments. As a professional reference substance, it can ensure the accuracy and repeatability of Lapatinib impurity detection results, providing a reliable basis for pharmaceutical quality research and quality control, and strongly supporting the quality control work in drug research, development, and production processes.
Application Fields:
Quality Control: As an impurity reference standard, it is used to establish and validate the detection methods for impurities in Lapatinib bulk drugs and formulations, ensuring that the sensitivity and specificity of the detection methods meet strict pharmaceutical quality standards and guaranteeing drug quality and safety.
Process Optimization: During the production of Lapatinib, by monitoring the content of this impurity and analyzing the reaction steps and stages of its generation, it helps to optimize the synthesis process, reduce the amount of impurity generation, and improve product quality and production efficiency.
Stability Studies: In drug stability tests, it explores the change rules of this impurity under different storage conditions (such as temperature, humidity, light), providing key data support for determining the shelf life and suitable storage conditions of drugs.
Background Description: Lapatinib is a key targeted drug for the treatment of HER2-positive breast cancer. In its research, development, production, and quality control processes, impurity research is a core link to ensure drug safety and effectiveness. The presence of impurities may interfere with the normal mechanism of the drug and even cause potential adverse reactions. As a related impurity of Lapatinib, in-depth research on Lapatinib Impurity 12 helps to comprehensively and accurately evaluate the quality of Lapatinib drugs, improve the drug quality evaluation system, and ensure the safety and effectiveness of clinical medication.
Research Status: Currently, research on Lapatinib Impurity 12 is continuously deepening. In terms of detection technology, researchers are actively exploring advanced detection methods such as Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry (UPLC-MS/MS) and high-resolution mass spectrometry to achieve high-precision detection of trace impurities. In the study of impurity generation mechanisms, the formation causes and influencing factors are deeply analyzed by simulating drug synthesis reactions and storage environments, providing a theoretical basis for controlling impurities from the source. In addition, research on the impact of this impurity on the efficacy and safety of Lapatinib is also gradually being carried out, aiming to further clarify its role in drug quality evaluation and provide more comprehensive theoretical support for rational clinical medication
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China


