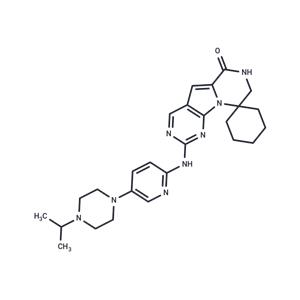
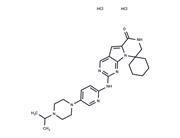
| Name | Lerociclib |
| Description | Lerociclib (G1T38) is a selective and highly potent CDK4/6 inhibitor with anticancer and antitumor activity, inhibits CDK4/CyclinD1 and CDK6/CyclinD3, and inhibits tumor growth in an animal model of endocrine-resistant breast cancer. Due to the poor solubility, for animal studies, the salt form T11345L is recommended. |
| In vitro | Lerociclib is highly potent and selective for CDK4/cyclin D1 and CDK6/cyclin D3 over CDK1, CDK2, CDK5 and CDK7 and their respective binding partners. Lerociclib produced a robust and sustained G1 arrest in CDK4/6 dependent cells with an EC50 of ~20 nmol/L. [1] |
| In vivo | To evaluate the murine PK/PD relationship of Lerociclib, an ER+ MCF7 xenograft model was utilized. Compound concentrations were measured in plasma and tumors from tumor-bearing mice given a single oral dose of Lerociclib (100 mg/kg). Oral Lerociclib has sufficient mouse PK/PD for assessing efficacy in tumor models. [1] |
| Storage | Store at low temperature,Keep away from moisture | Shipping with blue ice/Shipping at ambient temperature. |
| Keywords | Lerociclib | CDK9/Cyclin T | CDK7/Cyclin H/MAT1 | cdk6/cyclin D3 | CDK6 | CDK5/p35 | Cdk5/p25 | Cdk4/cyclin D1 | CDK4 | CDK2/cyclinE | cdk2/cyclin A | CDK1/cyclinB1 |
| Inhibitors Related | Ribociclib | (E)-β-Farnesene | Amantadine | 2-Chloropyrazine | Kojic acid | PF07104091 | Abemaciclib | Palbociclib | Abemaciclib methanesulfonate | Sodium Oxamate | Seliciclib | Dinaciclib |
| Related Compound Libraries | Anti-Lung Cancer Compound Library | Bioactive Compound Library | Kinase Inhibitor Library | Anti-Cancer Clinical Compound Library | Drug Repurposing Compound Library | Clinical Compound Library | Bioactive Compounds Library Max | Cell Cycle Compound Library | Anti-Cancer Compound Library | Anti-Cancer Drug Library | Anti-Cancer Active Compound Library | High-Efficiency Gene Editing Small Molecule Library |

 United States
United States