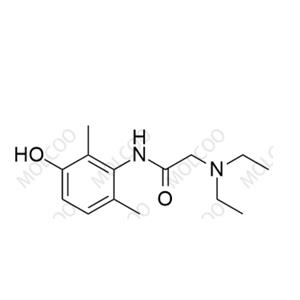
Product Number: L017053
English Name: Lidocaine Impurity 53
English Alias: 2-(diethylamino)-N-(3-hydroxy-2,6-dimethylphenyl)acetamide
CAS Number: 34604-55-2
Molecular Formula: C₁₄H₂₂N₂O₂
Molecular Weight: 250.34
As an impurity reference standard for lidocaine, this compound has a well-defined structure and stable properties, serving as a standard substance for drug research, development, and quality control. Its advantages include:
Enabling precise analysis of the side reaction mechanism of hydroxyl substitution positional isomerization during lidocaine synthesis to optimize processes and reduce the generation of 3-hydroxy impurities;
As a polar impurity reference standard containing hydroxyl and acetamide groups, improving the separation and quantitative accuracy of HPLC and other detection methods for positional isomeric impurities;
Helping study the impact of hydroxyl position changes on drug stability and toxicological properties to provide a scientific basis for impurity control strategies.
Drug Development: Used as an impurity reference standard to identify and quantify 3-hydroxy isomeric impurities in lidocaine and its formulations, evaluating the purity of APIs and formulations;
Quality Control: Acting as a standard substance to validate the specificity of detection methods (e.g., HPLC, LC-MS), ensuring the impurity content meets pharmacopoeia requirements during production;
Process Optimization: Guiding the optimization of phenolic hydroxyl substitution reaction conditions (such as temperature, catalyst) in the synthesis route by analyzing the formation path of this impurity to reduce impurity generation.
Lidocaine is a commonly used local anesthetic in clinical practice. If the phenolic hydroxyl position in its molecular structure undergoes isomerization (such as 3-hydroxy substitution), Lidocaine Impurity 53 is easily generated. In drug synthesis, insufficient raw material purity or out-of-control electrophilic substitution reaction conditions may lead to the production of this impurity. Since positional isomeric impurities may affect the pharmacological activity and safety of drugs, research on them is an important part of the lidocaine quality control system.
Current research focuses on:
Detection Method Optimization: Using HPLC-UV or LC-MS combined techniques to improve the resolution of 3-hydroxy and 4-hydroxy impurities by optimizing chromatographic conditions (such as mobile phase polarity, column temperature);
Synthesis Mechanism Research: Tracking the regioselectivity of hydroxyl substitution reactions through isotope labeling to develop highly regioselective synthesis processes;
Toxicological Evaluation: Comparing the safety differences between this impurity and lidocaine through in vitro cytotoxicity experiments to provide data support for setting impurity limits;
Stability Studies: Investigating the degradation behavior of this impurity under acidic/alkaline conditions to evaluate its impact on the stability of lidocaine injections.





 China
China