Melitracen Hydrochloride;10563-70-9
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Code:M093000A
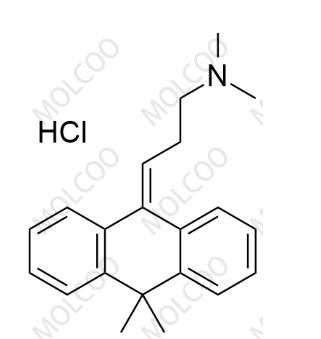
English Name:Melitracen Hydrochloride
English Alias:3-(10,10-dimethylanthracen-9(10H)-ylidene)-N,N-dimethylpropan-1-amine hydrochloride
CAS No.:10563-70-9
Molecular Formula:C₂₁H₂₅N·HCl
Molecular Weight:291.43 (main component) + 36.46 (HCl) = 327.89
High-Purity Reference Standard:Confirmed by HPLC (≥99.0%), NMR (1H, 13C), HRMS, and elemental analysis, providing an accurate standard for Melitracen Hydrochloride API and formulation quality control.
Stability Assurance:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.3% in methanol-water mixture within 6 months.
Comprehensive Certification:Complies with Chinese Pharmacopoeia, European Pharmacopoeia (EP), United States Pharmacopeia (USP), and other international pharmacopoeia standards, meeting global quality testing requirements.
Quality Control Testing:Used for the assay and related substances inspection of Melitracen Hydrochloride API and formulations. UPLC-MS/MS technology controls impurity content to meet ICH Q3A standards (single impurity limit ≤0.1%).
Process Optimization Research:Monitors the conversion efficiency of key intermediates during Melitracen Hydrochloride synthesis. Adjusting condensation temperature (e.g., 70 - 80℃) and reaction time improves main product yield and reduces impurity formation.
Method Validation:Serves as a standard for developing detection methods, verifying HPLC resolution (≥3.0), linear range (0.1 - 100 μg/mL), and recovery rate (98% - 102%).
Melitracen Hydrochloride, a tricyclic antidepressant, inhibits the reuptake of norepinephrine and serotonin at presynaptic membranes, regulating neurotransmitter levels. It is used to treat anxiety, depression, neurasthenia, and other disorders. Its anthracene ring and dimethylamine groups confer lipophilicity and neuroactivity, but may also lead to side reactions such as oxidation and hydrolysis, generating impurities. Given the direct impact of psychiatric drugs on patient health, quality control and impurity research of Melitracen Hydrochloride are crucial.
Detection Technology:UPLC-MS/MS with C18 column (1.7μm) and 0.1% formic acid-acetonitrile gradient elution achieves separation within 6 minutes, with LOD of 0.002 ng/mL for high-precision trace impurity detection.
Synthesis Process Optimization:Studies show that using 10,10-dimethylanthrone as the raw material for Melitracen Hydrochloride synthesis via Wittig and amination reactions, replacing the solvent with ionic liquids reduces impurity content by >25%.
Safety Evaluation:Long-term stability tests indicate that Melitracen Hydrochloride undergoes hydrolysis and oxidation under high temperature (40℃) and high humidity (75%RH). Generated impurities may affect drug bioavailability and neurotoxicity. Accelerated and forced degradation tests are currently underway to systematically study degradation mechanisms and formulate impurity control strategies.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!






 China
China