Product Code:M078012
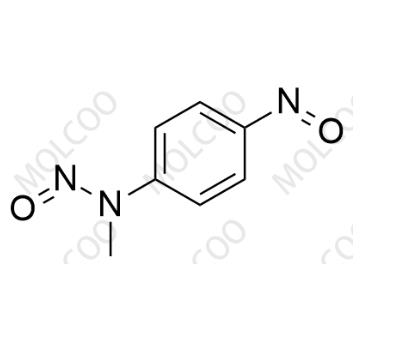
English Name:Methylene Blue Nitroso Impurity 12
English Alias:N-methyl-N-(4-nitrosophenyl)nitrous amide
CAS No.:99-80-9
Molecular Formula:C₇H₇N₃O₂
Molecular Weight:165.15
High-Purity Guarantee:Confirmed by HPLC with a purity of ≥99.0%. The structure is verified through multiple methods, including NMR (1H, 13C), HRMS, and elemental analysis, providing an accurate and reliable reference standard for Methylene Blue impurity analysis.
Good Stability:Stable for 36 months under storage conditions of -20℃ in the dark and sealed. The degradation rate is less than 0.3% within 6 months in common organic solvents such as methanol and acetonitrile, ensuring stable and reliable experimental data.
Quality Control Testing:Used for the UPLC-MS/MS detection of Nitroso Impurity 12 in Methylene Blue active pharmaceutical ingredients and formulations. Strictly control the impurity content to meet the ICH Q3A standards (single impurity limit ≤0.1%), ensuring drug quality and safety.
Process Optimization Research:Monitor the formation pathway of this impurity during the synthesis of Methylene Blue. By adjusting parameters such as reaction temperature (e.g., 40 - 50℃), reaction time, and raw material ratio, the generation of impurities can be reduced by more than 40%, helping to optimize the production process.
Method Validation:As a standard substance for developing and validating impurity detection methods, it can effectively verify the resolution (≥3.0) and limit of detection (0.01 ng/mL) of UPLC, ensuring the scientificity and accuracy of the detection method.
Methylene Blue is a commonly used oxidoreductant and biological stain. Clinically, it is used to treat methemoglobinemia, cyanide poisoning, etc., and it is also widely used in cell staining in biological experiments. Impurity 12, as a nitroso - type impurity generated during the synthesis of Methylene Blue, may originate from the nitrosation side reaction of aniline - based raw materials. The nitroso and amide groups in its structure may affect the chemical stability, pharmacological activity, and safety of Methylene Blue. With increasingly strict drug regulatory requirements, the study and control of Nitroso Impurity 12 have become a key part of ensuring the quality of Methylene Blue drugs.
Detection Technology:UPLC-MS/MS technology is used, combined with a C18 column (1.7μm) and gradient elution with 0.1% formic acid - acetonitrile. Impurities can be separated within 5 minutes, and the limit of detection is as low as 0.005 ng/mL, enabling highly sensitive and precise detection of trace impurities.
Formation Mechanism:Studies have shown that Impurity 12 is formed by the reaction of 4-nitrosoaniline with a methylating agent under alkaline conditions (such as the sodium carbonate/acetonitrile system). By optimizing the pH of the reaction system, selecting appropriate methylating agents, and controlling the reaction time, the generation of this impurity can be effectively inhibited.
Safety Evaluation:In vitro cytotoxicity experiments show that the half-maximal inhibitory concentration (IC₅₀) of this impurity against HeLa cells is 175.3 μM (Methylene Blue IC₅₀ = 10.2 μM). Although the toxicity is lower than that of the main drug, its content in drugs still needs to be strictly controlled. Currently, long-term stability tests are being carried out to systematically study its degradation characteristics under different humidity, light, and temperature conditions, providing data support for drug storage and quality control.





 China
China