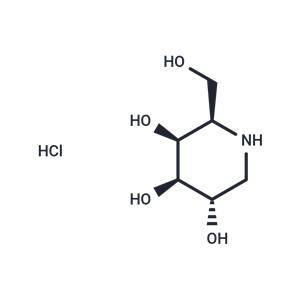
| Name | Migalastat hydrochloride |
| Description | Migalastat hydrochloride (GR181413A) is an orally available, potent and competitive inhibitor of alpha-galactosidase A. It promotes the transport of alpha-galactosidase A to the lysosome and can be used in the study of Fabry disease. |
| In vitro | Migalastat hydrochloride (GR181413A) demonstrates IC50 and Ki values of 0.04 μM for human lysosomal alpha-Gal A[4]. |
| In vivo | Fabry disease is an X-linked recessive disorder caused by deficient activity of alpha-galactosidase A (α-Gal A)[2]. In transgenic mice expressing the mutant human α-Gal A (TgM), oral administration of Migalastat hydrochloride (3 mg/kg per day for 4 consecutive weeks) results in a dose- and time-dependent increase in α-Gal A activity in the heart, kidneys, spleen, and liver[2].After a 2-week pretreatment with Migalastat hydrochloride, the half-life for all major issues is less than 1 day[2]. Administration of Migalastat hydrochloride (100 mg/kg per day orally for 28 days) in transgenic mice leads to a reduction of 64%, 59%, and 81% in globotriaosylceramide (Gb3) levels in the kidneys, heart, and skin, respectively[3]. |
| Storage | Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | H2O : 50 mg/mL (250.46 mM), Sonication is recommended.
|
| Keywords | α-galactosidase A | Migalastat hydrochloride | GR181413A |
| Inhibitors Related | Cedryl acetate | X-GAL | Hexylresorcinol | Conduritol B epoxide | Gcase activator 2 | NCGC00092410 | Fucoidan | Miglitol | Ambroxol hydrochloride | D-(-)-Quinic acid | Naringinase | Acarbose |
| Related Compound Libraries | FDA-Approved & Pharmacopeia Drug Library | Bioactive Compound Library | Approved Drug Library | EMA Approved Drug Library | Toxic Compound Library | Drug Repurposing Compound Library | Inhibitor Library | FDA-Approved Drug Library | Orally Active Compound Library | Bioactive Compounds Library Max |

 United States
United States