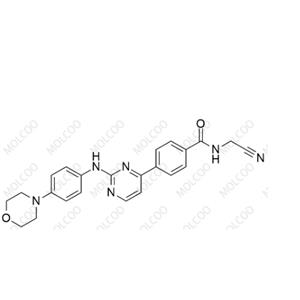
Product Number: M094000
English Name: Momelotinib
English Alias: N-(cyanomethyl)-4-(2-((4-morpholinophenyl)amino)pyrimidin-4-yl)benzamide
CAS Number: 1056634-68-4
Molecular Formula: C₂₃H₂₂N₆O₂
Molecular Weight: 414.46
As a dual JAK1/2 inhibitor, it shows high selectivity for the Janus kinase family, precisely inhibiting the JAK-STAT signaling pathway to reduce off-target effects;
The structure contains cyano, morpholine ring, and pyrimidine amine groups, exhibiting favorable pharmacokinetic properties in vitro and in vivo (e.g., high oral bioavailability, moderate half-life);
Strong chemical stability, suitable for the development of various dosage forms, with clear impurity control standards meeting the quality requirements of new drug research and development.
Clinical Research: Used for treating hematological diseases associated with abnormal JAK pathway such as myelofibrosis and polycythemia vera, currently in the clinical trial stage;
Basic Research: As a JAK1/2 inhibitor tool compound, used to explore the mechanism of the JAK-STAT signaling pathway in immune regulation and cell proliferation;
Drug Development: Can be used as a lead compound for structure optimization or to develop combination therapies to enhance anti-tumor and anti-inflammatory effects.
Momelotinib is an oral small-molecule JAK1/2 inhibitor developed by GlaxoSmithKline (GSK). Its mechanism of action inhibits the activity of JAK1 and JAK2 kinases, blocking STAT protein phosphorylation, thereby inhibiting abnormal cell proliferation and inflammatory signal transduction. In diseases such as myelofibrosis, the JAK-STAT pathway is overactivated, and Momelotinib demonstrates therapeutic potential by targeting this pathway, with its safety and efficacy evaluated in multiple clinical trials.
Clinical Trial Progress: Completed phase III clinical trials (such as SIMIPLIFY-1 and SIMIPLIFY-2) for treating myelofibrosis, showing improvement in patient anemia symptoms and spleen volume;
Combination Therapy Research: Exploring the feasibility of combining with other targeted drugs (such as BCL-2 inhibitors) for hematological tumor treatment;
Drug Resistance Mechanism Research: Analyzing the resistance mechanism of JAK2 V617F mutant patients after long-term medication to guide subsequent drug optimization;
Safety Evaluation: Focusing on adverse reactions such as anemia and thrombocytopenia during treatment, optimizing the dosage regimen to balance efficacy and safety.





 China
China