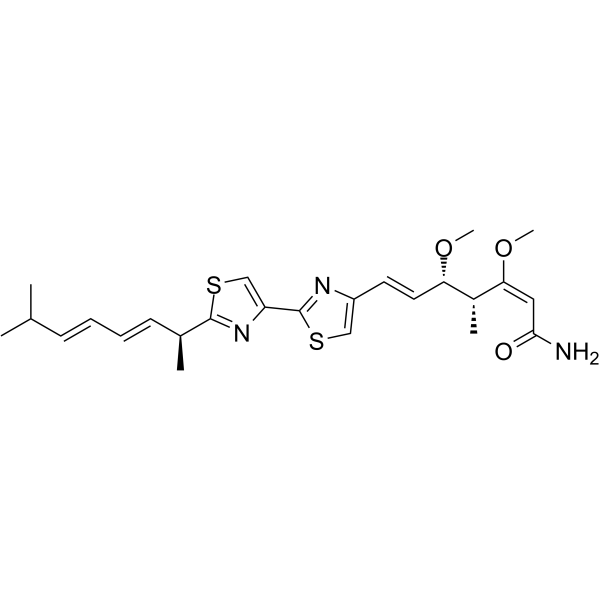
PRODUCT INFORMATION
| Common Name | Myxothiazol |
|---|
| CAS Number | 76706-55-3 | Molecular Weight | 487.67800 |
|---|
| Density | 1.158g/cm3 | Boiling Point | 679.6ºC at 760 mmHg |
|---|
| Molecular Formula | C25H33N3O3S2 | Melting Point | N/A |
|---|
| MSDS | ChineseUSA | Flash Point | 364.8ºC |
|---|
| Symbol | 
GHS06 | Signal Word | Danger |
|---|
Use of Myxothiazol
Myxothiazol, an antifungal antibiotic, is a mitochondrial electron transport chain complex III (bc1 complex) inhibitor. Myxothiazol inhibits the growth of many yeasts and fungi at concentrations between 0.01 and 3 μg/ml[1][2]. |
Properties
Articles39
Spectrum
Names
| Name | myxothiazol |
|---|
| Synonym | More Synonyms |
|---|
Myxothiazol Biological Activity
| Description | Myxothiazol, an antifungal antibiotic, is a mitochondrial electron transport chain complex III (bc1 complex) inhibitor. Myxothiazol inhibits the growth of many yeasts and fungi at concentrations between 0.01 and 3 μg/ml[1][2]. |
|---|
| Related Catalog | Research Areas >> Infection Signaling Pathways >> Anti-infection >> Fungal |
|---|
| In Vitro | Myxothiazol inhibits the growth of many yeasts and fungi at concentrations between 0.01 and 3 μg/ml[2]. Myxothiazol binds to the ubiquinol oxidation site Qo of complex III and blocks electron transfer from ubiquinol to cytochrome b and thus inhibits complex III activity[3]. |
|---|
| In Vivo | Myxothiazol (i.p.; 0.56 mg/kg; daily for 4 days)-induced complex III inhibition can be induced in mice for four days in a row without overt hepatotoxicity or lethality[3]. Animal Model: C57Bl/J6 mice[3] Dosage: 0.56 mg/kg Administration: I.p.; 24 hours intervals for at most 4 times Result: A reversible complex III activity decrease to 50% of control value occurred at 2 h post-injection. At 74 h only minor histological changes in the liver were found, supercomplex formation was preserved and no significant changes in the expression of genes indicating hepatotoxicity or inflammation were found. |
|---|
| References | [1]. Thierbach G, et al. Myxothiazol, a new antibiotic interfering with respiration. Antimicrob Agents Chemother. 1981;19(4):504-507. [2]. von Jagow G,et al. An inhibitor of mitochondrial respiration which binds to cytochrome b and displaces quinone from the iron-sulfur protein of the cytochrome bc1 complex. J Biol Chem. 1984;259(10):6318-6326. [3]. Davoudi M, et al. A mouse model of mitochondrial complex III dysfunction induced by myxothiazol. Biochem Biophys Res Commun. 2014;446(4):1079-1084. |
|---|
Chemical & Physical Properties
| Density | 1.158g/cm3 |
|---|
| Boiling Point | 679.6ºC at 760 mmHg |
|---|
| Molecular Formula | C25H33N3O3S2 |
|---|
| Molecular Weight | 487.67800 |
|---|
| Flash Point | 364.8ºC |
|---|
| Exact Mass | 487.19600 |
|---|
| PSA | 143.81000 |
|---|
| LogP | 6.51870 |
|---|
| Index of Refraction | 1.584 |
|---|
| Storage condition | -20°C |
|---|
Toxicological Information
CHEMICAL IDENTIFICATIONCHEMICAL NAME : 2,6-Heptadienamide, 7-(2'-(1,6-dimethyl-2,4-heptadienyl)(2,4'-bithiazol)- 4-yl)-3,5-dimethoxy- 4-methyl-
CAS REGISTRY NUMBER : 76706-55-3
MOLECULAR FORMULA : C25-H33-N3-O3-S2
HEALTH HAZARD DATAACUTE TOXICITY DATAROUTE OF EXPOSURE : Unreported
SPECIES OBSERVED : Rodent - mouse
REFERENCE : JANTAJ Journal of Antibiotics. (Japan Antibiotics Research Assoc., 2-20-8 Kamiosaki, Shinagawa-ku, Tokyo, 141, Japan) V.2-5, 1948-52; V.21- 1968- Volume(issue)/page/year: 33,1474,1980
|
Safety Information
| Symbol | 
GHS06 |
|---|
| Signal Word | Danger |
|---|
| Hazard Statements | H300 |
|---|
| Precautionary Statements | P264-P301 + P310 |
|---|
| Hazard Codes | T+ |
|---|
| Risk Phrases | 28 |
|---|
| Safety Phrases | 28-36/37-45 |
|---|
| RIDADR | UN 3462 6.1/PG 1 |
|---|
| Packaging Group | III |
|---|
| Hazard Class | 6.1(b) |
|---|
Company Profile
Our company is a high-tech enterprise specializing in chemical raw materials such as electronic materials, optoelectronic materials, semiconductor materials, UV monomers, silane catalysts, cosmetic raw materials, etc. The company integrates research and development, production, customized synthesis, and sales, and is committed to providing customers with high-quality chemical product solutions.
Our Company


Our Advantage
Rich experience
Our products have been exported to many countries, including Germany, Spain, the United Kingdom, the United States, Australia, the Middle East, Asia, and more. We have received highly positive feedback from our clients and have established long-term friendly cooperative relationships with them.
Excellent quality, purity, and competitive price
Excellent quality is one of the cornerstones of our success. We welcome ordering samples for quality testing.

Safe and fast delivery
We will arrange shipment of spot products as soon as we receive payment.
Customized products are determined based on the synthesis time of different products.
Excellent pre-sales and after-sales service
Pre sales:
We are committed to providing the most favorable quotes and detailed information about our products and company.
after-sale service:
We assist buyers in customs clearance by providing necessary documents and information.
If there is any dispute over product quality, we are committed to providing the best solution.
• Packaging:
1kg/bag, 25kg/drum, 50kg/drum, 180kg/drum, 200kg/drum, 1T/bag/drum, standard export packaging or packaging required by customers


• Shipping

• Delivery:
Within 7 days after receiving your payment
Contact information
For more details, pls contact us freely.
Contact name: Tina
Email address: Tina@fdachem.com
Mob: 86 13213167925
WhatsApp/Skype/Wechat/LINE: 86 15225627621

 China
China
![4-(2''-benzothiazolyl)sulfonylmethyl-2'-[(1'''S),6'''-dimethylhepta-(2'''E),(4'''E)-dienyl]-2,4'-bithiazole structure](https://image.chemsrc.com/caspic/398/1093126-36-3.png)