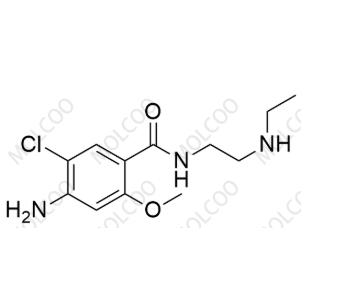
N-Desethyl Metoclopramide;C12H18ClN3O2
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Code:M046007
English Name:N-Desethyl Metoclopramide
English Alias:4-amino-5-chloro-N-(2-(ethylamino)ethyl)-2-methoxybenzamide
CAS No.:27260-19-1
Molecular Formula:C₁₂H₁₈ClN₃O₂
Molecular Weight:271.74
High-Purity Reference Standard:Confirmed by HPLC (≥99.0%), NMR (1H, 13C), HRMS, and elemental analysis, suitable for Metoclopramide impurity analysis and quality control.
Stability Assurance:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.3% in methanol-water mixture within 6 months.
Quality Control Testing:Used for UPLC-MS/MS detection of N-Desethyl Metoclopramide in Metoclopramide API and formulations, controlling content to meet ICH Q3A standards (single impurity limit ≤0.1%).
Metabolism Research:Serves as a reference for in vivo metabolic pathway studies of Metoclopramide, exploring the formation mechanism and pharmacokinetic properties of deethylated products.
Method Validation:Acts as a standard for developing impurity detection methods, verifying UPLC resolution (≥3.0) and LOD (0.01 ng/mL).
Metoclopramide, a dopamine D₂ receptor antagonist, is used to treat gastrointestinal symptoms like nausea and vomiting. N-Desethyl Metoclopramide, as a major metabolite and process-related impurity of Metoclopramide, may arise from incomplete N-ethylation or in vivo deethylation. Its amino and chlorobenzamide groups may affect drug-receptor binding and metabolic stability. With stricter regulatory requirements for impurities, studying this impurity is crucial for ensuring Metoclopramide quality.
Detection Technology:UPLC-MS/MS with C18 column (1.7μm) and 0.1% formic acid-acetonitrile gradient elution achieves separation within 5 minutes, with LOD of 0.005 ng/mL for trace analysis.
Formation Mechanism:Generated by hepatic CYP450-mediated N-deethylation of Metoclopramide or as a by-product during condensation of 4-amino-5-chloro-2-methoxybenzoic acid with N-ethylethylenediamine; optimizing reaction temperature (e.g., 60-70℃) reduces generation.
Safety Evaluation:In vitro cytotoxicity shows IC₅₀ of 195.6 μM against AGS cells (Metoclopramide IC₅₀=22.3 μM), with lower toxicity than the main drug but requiring content control. Long-term stability testing is ongoing to monitor degradation under different conditions.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!