N-Nitroso Benazepril
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Number: N031054
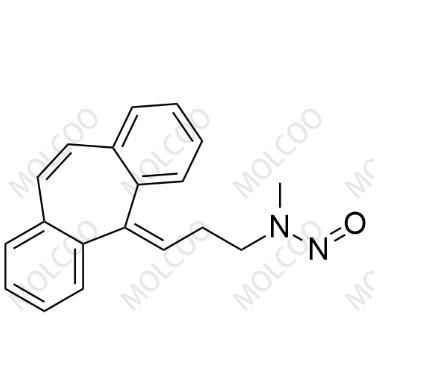
English Name: N-Nitroso Desmethyl Cyclobenzaprine
English Alias: N-(3-(5H-dibenzo[a,d][7]annulen-5-ylidene)propyl)-N-methylnitrous amide
CAS Number: None
Molecular Formula: C₁₉H₁₈N₂O
Molecular Weight: 290.36
As a nitroso derivative impurity of Desmethyl Cyclobenzaprine, this compound has the following advantages:
Well-defined and distinct structure: Contains nitroso (-N=O) group, dibenzoheptalene skeleton, and propyl chain, differing from Desmethyl Cyclobenzaprine by nitroso substitution on the amino group. It can be accurately identified by techniques such as HPLC and GC-MS, providing a specific marker for impurity detection;
High stability and traceability: The N-nitrosamine structure is relatively stable under dark conditions. As a product of further oxidation of cyclobenzaprine after in vivo demethylation, it directly reflects the degree of metabolic transformation, improving the accuracy of quality and metabolic tracing;
High detection sensitivity: The conjugated system of nitroso group shows characteristic absorption in the UV region (230-250nm), and combined with the strong hydrophobicity of dibenzoheptalene, trace analysis can be achieved via HPLC-UV or GC-MS, compatible with detection systems for lipophilic drugs.
Pharmaceutical quality control: Used as an impurity reference standard to identify and quantify N-Nitroso Desmethyl Cyclobenzaprine in cyclobenzaprine APIs and formulations, ensuring residual nitroso impurities from metabolism or storage meet quality standards;
Metabolic pathway research: Exploring the demethylation-nitrosation metabolic pathway of cyclobenzaprine in vivo, analyzing the formation and clearance rules of this impurity to provide data support for drug safety evaluation;
Stability assessment: Monitoring the content of this impurity to evaluate the oxidative deterioration trend of cyclobenzaprine formulations during long-term storage, providing reference for formulating reasonable storage conditions.
Cyclobenzaprine is a central muscle relaxant used to relieve muscle spasms, with a structure containing a tertiary amine group. After in vivo demethylation catalyzed by CYP450 enzymes, the generated Desmethyl Cyclobenzaprine may be further oxidized to form N-(3-(5H-dibenzo[a,d][7]annulen-5-ylidene)propyl)-N-methylnitrous amide (i.e., N-Nitroso Desmethyl Cyclobenzaprine). N-nitrosamines have potential genotoxicity, making their control an important part of cyclobenzaprine quality assurance and safety evaluation.
Current research focuses on:
Detection method optimization: Using UPLC-MS/MS with optimized parameters based on characteristic fragment ions of nitroso groups (e.g., m/z 46 [NO]⁺) to achieve trace detection of this impurity (detection limits up to ppb level);
Metabolic mechanism analysis: Identifying key enzymes (e.g., CYP2E1) catalyzing nitrosation of Desmethyl Cyclobenzaprine through in vitro liver microsome experiments, clarifying the biosynthetic pathway of this impurity;
Toxicity assessment: Evaluating the potential hazards of this nitroso derivative through in vitro cell genotoxicity tests (e.g., chromosome aberration test) to provide a scientific basis for formulating strict impurity limits;
Inhibition strategy research: Developing additives targeting nitrosation inhibition (e.g., ascorbic acid derivatives) to reduce the formation of this impurity in cyclobenzaprine formulations during storage.
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com