Product Code:S027038
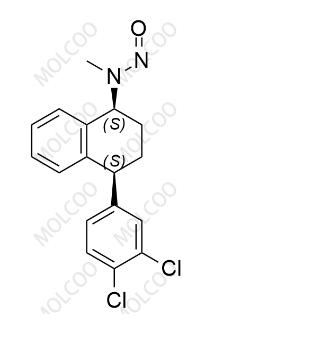
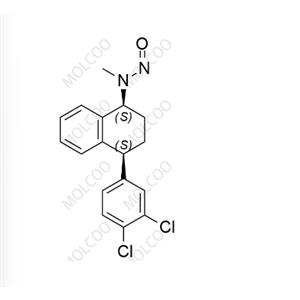
English Name:N-Nitroso Sertraline
English Alias:N-((1S,4S)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydronaphthalen-1-yl)-N-methylnitrous amide
CAS No.:3006789-98-3
Molecular Formula:C₁₇H₁₆Cl₂N₂O
Molecular Weight:335.23
High-Purity Reference Standard:Confirmed by HPLC (≥99.0%), NMR, and HRMS, suitable for precise analysis of nitrosamine impurities in Sertraline.
Stability Assurance:Stable for 24 months at -20℃ under light-protected, sealed storage; degradation rate <0.5% in acetonitrile solution within 3 months.
Quality Control Testing:Used for LC-MS/MS detection of N-nitroso impurities in Sertraline API and formulations, controlling impurity content to meet ICH M7 standards (≤0.1 ppm).
Process Optimization Research:Monitors N-Nitroso Sertraline generated from nitrosation side reactions during Sertraline synthesis, reducing formation by adjusting reaction temperature (e.g., 0-5℃) and pH (weak acidic conditions).
Method Validation:Serves as a standard for developing nitrosamine detection methods, verifying UPLC resolution (≥2.0) and LOD (0.005 ng/mL).
Sertraline, a selective serotonin reuptake inhibitor (SSRI), is used in the treatment of depression and other mental illnesses. Nitrosamine impurities (such as N-Nitroso Sertraline) pose potential genotoxic and carcinogenic risks, possibly formed by reactions between secondary amines and nitrosating agents during drug synthesis. With stricter control requirements from international regulatory agencies (FDA, EMA), studying such impurities has become crucial for ensuring Sertraline safety.
Detection Technology:UPLC-MS/MS with C18 column (1.7μm) and 0.1% formic acid-acetonitrile gradient elution achieves separation within 2 minutes, with LOD of 0.003 ng/mL for trace analysis.
Formation Mechanism:Originates from nitrosation of the amine group in Sertraline, with higher reactivity under acidic conditions (pH 2-3). Adding nitrosamine inhibitors (e.g., vitamin C) or optimizing purification (e.g., column chromatography) reduces content by >70%.
Safety Evaluation:Preliminary toxicology shows positive Ames test results, indicating potential mutagenicity. Current standards limit it to ≤0.1 ppm, requiring continuous monitoring via accelerated and long-term stability studies.
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China