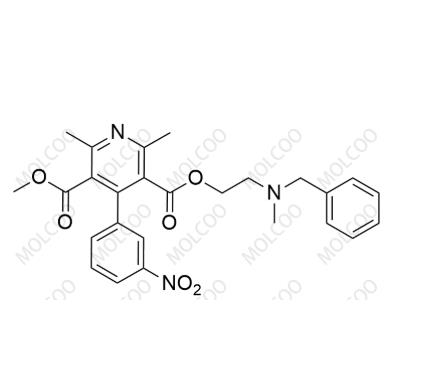
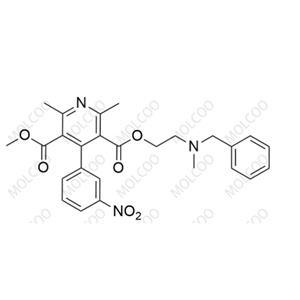
Product Number: N020013
English Name: Nicardipine EP Impurity A
English Alias: 3-(2-(benzyl(methyl)amino)ethyl) 5-methyl 2,6-dimethyl-4-(3-nitrophenyl)pyridine-3,5-dicarboxylate
CAS Number: 59875-58-0
Molecular Formula: C₂₆H₂₇N₃O₆
Molecular Weight: 477.51
As a European Pharmacopoeia (EP) standard impurity of nicardipine, the research advantages of this compound include:
Serving as an EP-compliant reference standard for direct use in quality testing of nicardipine APIs and formulations, ensuring consistency with international pharmacopoeia requirements;
Helping to analyze the by-product formation mechanism of amino substitution reactions in nicardipine synthesis to optimize processes and reduce the formation of benzylmethylamine impurities;
Assisting in evaluating the impact of nitro and amino structures on drug stability and safety to provide a basis for establishing storage conditions and quality standards.
Quality Control: Used as an EP standard impurity reference for system suitability tests in HPLC and other detection methods to verify whether the content of this impurity in nicardipine meets EP limits;
Drug Development: In generic drug research, used to compare the impurity profile of the original drug to ensure quality consistency between the generic and original drug;
Process Optimization: Guiding the optimization of amination and pyridine ring condensation reaction conditions in the synthesis route to reduce the generation of this impurity through impurity content analysis.
Nicardipine is a commonly used dihydropyridine calcium channel blocker in clinical practice for the treatment of hypertension and angina. If amino substitution reactions or pyridine ring construction are not properly controlled during its synthesis, impurities containing benzylmethylamine structures (such as Impurity A) are easily generated. The European Pharmacopoeia (EP) has clear limits for this impurity, making research on it a necessary part of international quality control for nicardipine.
Current research focuses on:
Detection Method Optimization: Using EP-recommended HPLC-UV methods or LC-MS combined techniques to improve the detection sensitivity and specificity of this impurity;
Synthesis Process Improvement: Reducing the generation of benzylmethylamine derivatives by adjusting the reagent ratio and temperature of amination reactions;
Stability Studies: Investigating the degradation behavior of this impurity under light, high temperature, and high humidity conditions to evaluate its impact on nicardipine formulation stability;
Toxicological Evaluation: Studying the potential genotoxicity of nitro and amino structures through in vitro cell experiments to provide data support for establishing safe limits.





 China
China