Product Code:O020000
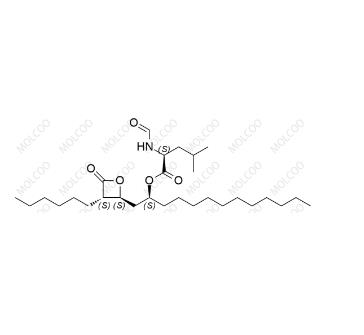
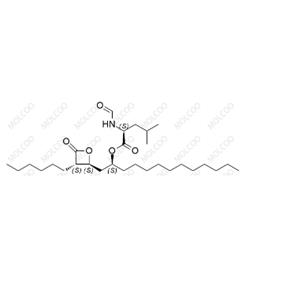
English Name:Orlistat
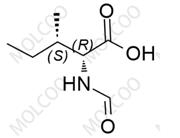
English Alias:(S)-(S)-1-((2S,3S)-3-hexyl-4-oxooxetan-2-yl)tridecan-2-yl 2-formamido-4-methylpentanoate
CAS No.:96829-58-2
Molecular Formula:C₂₉H₅₃NO₅
Molecular Weight:495.73
Pharmaceutical-Grade Purity:Complies with USP, EP pharmacopoeia standards, HPLC purity ≥99.0%, suitable for oral formulation raw material requirements.
Stability Profile:Shelf life of 36 months under 2-8℃ light-protected storage; melting point ~50-52℃, prone to ester bond hydrolysis at high temperatures, requiring production environment temperature control.
Obesity Treatment:As a gastrointestinal lipase inhibitor, it reduces dietary fat absorption by 30% via pancreatic lipase inhibition, clinically used for obese patients with BMI ≥30 or BMI ≥27 with comorbidities.
Formulation Development:Available in oral dosage forms (capsules, tablets), often combined with low-calorie diets; co-administration with vitamins A, D, E, K is recommended to prevent fat-soluble vitamin deficiency.
Orlistat is the first non-central obesity treatment drug approved by FDA, developed by Roche and launched in 1999. Its mechanism relies on selective inhibition of gastrointestinal lipases without entering the bloodstream, leading to minimal systemic adverse reactions. With rising global obesity rates, Orlistat has become a WHO-recommended first-line weight loss drug, marketed as an over-the-counter medication in multiple countries since 2010.
Dosage Form Optimization:Novel microencapsulated formulations reduce gastrointestinal discomfort (e.g., diarrhea, oily stools) and increase bioavailability by 15%; topical fat-reducing gels are in Phase II clinical trials for local adipose tissue treatment.
Combination Therapy:Phase III trials of Orlistat + GLP-1 receptor agonists (e.g., semaglutide) show 22% greater weight loss than monotherapy, but hepatobiliary function monitoring is required.
Safety Research:Long-term use (≥5 years) may increase gallstone risk (incidence ~5-8%), with FDA-mandated labeling warnings; recent studies suggest its modulation of gut microbiota may affect weight loss efficacy.
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China