Product No.:P055024
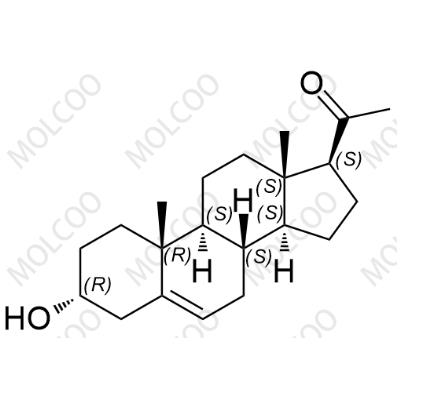
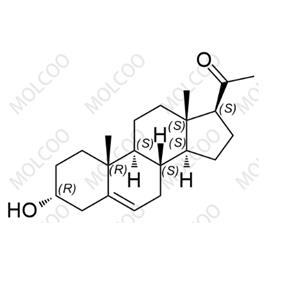
English Name:Progesterone Impurity 24
English Alias:1-((3R,8S,9S,10R,13S,14S,17S)-3-hydroxy-10,13-dimethyl-2,3,4,7,8,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl)ethanone
CAS No.:19037-28-6
Molecular Formula:C₂₁H₃₂O₂
Molecular Weight:316.48
High Purity:As a reference standard for progesterone impurities, it meets the requirements of pharmaceutical analysis, ensuring the accuracy and reliability of test results.
Well-Defined Structure:The chemical structure is confirmed, making it suitable for qualitative and quantitative analysis of impurities, providing a standard reference for pharmaceutical quality control.
Pharmaceutical Quality Control:Used for the detection and analysis of impurities in progesterone bulk drugs and preparations to ensure drug safety and compliance.
Analytical Method Validation:Serves as a reference standard for the development and validation of chromatographic and spectroscopic analytical methods, evaluating the sensitivity and specificity of the methods.
Progesterone is an important progestogen drug used in the treatment of menstrual disorders, threatened abortion, and other diseases. During drug synthesis and storage, various impurities may be generated, which can affect the efficacy and safety of the drug. As a related impurity of progesterone, Progesterone Impurity 24 has a steroid skeleton similar to that of progesterone. The study of this impurity helps to deeply understand the synthesis pathway, degradation mechanism, and quality control points of the drug.
Currently, research on progesterone and its impurities mainly focuses on the separation and identification of impurities, toxicity assessment, and control strategies. Studies on Progesterone Impurity 24 are mostly applied to the establishment of pharmaceutical analysis methods, such as quantitative detection by high-performance liquid chromatography (HPLC), mass spectrometry (MS), and other technologies. In addition, some studies focus on the formation rules of this impurity in drug stability tests to optimize the production process and storage conditions of the drug. With the improvement of ICH guidelines for pharmaceutical impurity control, the research on such impurities has become increasingly important in new drug development and generic drug consistency evaluation.
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China