Product Number: R006055A
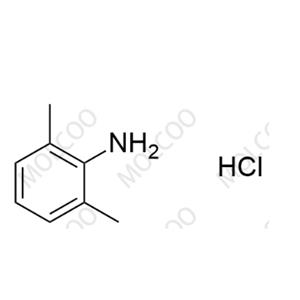
English Name: Ropivacaine EP Impurity H(Hydrochloride)
English Alias: 2,6-dimethylaniline hydrochloride
CAS Number: 21436-98-6
Molecular Formula: C₈H₁₁N·HCl
Molecular Weight: 157.64
As a European Pharmacopoeia (EP) standard impurity of ropivacaine, the research advantages of this compound include:
Analyzing the residual aniline raw materials or dealkylation side reaction mechanisms during ropivacaine synthesis to optimize processes for controlling aromatic amine impurity generation;
Serving as a hydrochloride-form reference standard to precisely match the actual state of drugs, improving the accuracy of impurity quantification;
Helping study the impact of aromatic amine structures on drug stability and toxicological properties (aromatic amines may have potential genotoxicity) to provide a scientific basis for impurity control.
Quality Control: Used as an EP standard impurity reference for system suitability tests in HPLC and other detection methods to verify whether the content of 2,6-dimethylaniline hydrochloride in ropivacaine meets EP limits;
Drug Development: In generic drug research, used to compare the impurity profile of the original drug to ensure quality consistency between the generic and original drug;
Process Optimization: Guiding the optimization of amination or deprotection reaction conditions in the synthesis route to reduce aromatic amine impurity generation through impurity content analysis.
Ropivacaine is a commonly used long-acting local anesthetic in clinical practice. If aniline raw materials (such as 2,6-dimethylaniline) remain or dealkylation reactions occur during its synthesis, this impurity is easily generated. The European Pharmacopoeia (EP) has clear limits for this impurity, and aromatic amines may have potential carcinogenic risks, making research on it a necessary part of international quality control for ropivacaine.
Current research focuses on:
Detection Method Optimization: Using EP-recommended HPLC-UV methods or LC-MS combined techniques to improve the detection sensitivity of this impurity (detection limit reaches ppm level);
Synthesis Process Improvement: Reducing the residue of 2,6-dimethylaniline or by-product generation by optimizing raw material ratios, reaction temperature, and other parameters;
Toxicological Evaluation: Studying the potential mutagenicity of this impurity through in vitro Ames tests and animal models;
Stability Studies: Investigating the degradation behavior of this impurity under light and high-temperature conditions to evaluate its impact on ropivacaine formulation stability





 China
China