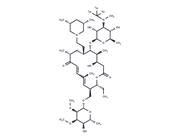
| Name | Tilmicosin |
| Description | Tilmicosin (EL-870) is a macrolide antibiotic. It is used in veterinary medicine for the treatment of bovine respiratory disease and ovine respiratory disease associated with Mannheimia haemolytica. |
| In vivo | After a subcutaneous injection, tilmicosin is eliminated more slowly from serum and milk, with t1/2 beta S of 29.3 and 41.4 hours, respectively. The apparent volume of distribution of tilmicosin is more than 1 L/kg. The peak serum tilmicosin concentration is 1.56 μg/mL. 6.39 hours after a subcutaneous injection of 10 mg/kg. Tilmicosin is extensively secreted into milk, reaching a maximum concentration of 11.6 μg/mL and having a large AUCmilk/AUCserum ratio of approximately 12:1. Tilmicosin is detectable in milk for 11 days after a single subcutaneous dose.[2] |
| Storage | Store at low temperature | Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | H2O : 8 mg/mL (9.2 mM), Sonication is recommended.
10% DMSO+40% PEG300+5% Tween 80+45% Saline : 2 mg/mL (2.3 mM), Sonication is recommended.
DMSO : 250 mg/mL (287.64 mM), Sonication is recommended.
Ethanol : < 1 mg/mL (insoluble or slightly soluble)
|
| Keywords | Tilmicosin | LY177370 | LY 177370 | Inhibitor | inhibit | EL870 | EL 870 | Bacterial | Antibiotic |
| Inhibitors Related | Neomycin sulfate | Aceglutamide | 2,4,6-Tri-tert-butylphenol | Adipic dihydrazide | D(+)-Raffinose pentahydrate | Sulfamethoxazole sodium | Terbinafine hydrochloride | Formamide | Hyaluronic acid sodium (MW 20 kDa) | Dimethyl sulfoxide | Sodium diacetate | Sodium bicarbonate |
| Related Compound Libraries | FDA-Approved & Pharmacopeia Drug Library | Macrocyclic Compound Library | Bioactive Compound Library | ReFRAME Related Library | Calcium Channel Compound Library | Anti-Gram-Negative Bacterial Library | Antibiotics Library | Anti-Bacterial Compound Library | NO PAINS Compound Library | Bioactive Compounds Library Max | Anti-Infection Compound Library | Human Metabolite Library |

 United States
United States