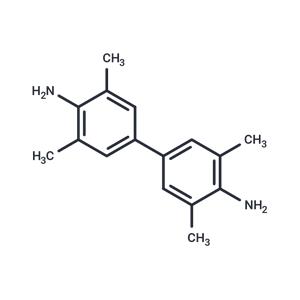
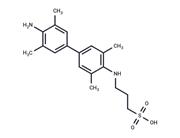
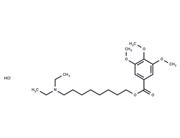
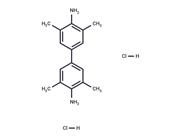
| In vitro | Instructions
I. Solution preparation
1. TMB substrate solution: Sure Blue TMB is usually provided as a concentrated solution and needs to be diluted according to the experimental requirements when used. The standard concentration of the substrate solution is 1x, and the concentrated solution is usually diluted proportionally with an appropriate solvent (such as deionized water). Before use, please refer to the manufacturer's instructions for preparation.
2. Hydrogen peroxide solution: The reaction of TMB usually requires hydrogen peroxide as an oxidant. The concentration of hydrogen peroxide solution is usually 0.01%-0.03%. Too high or too low concentrations may affect the sensitivity of the reaction.
II. Immunohistochemistry (IHC) application
1. Antibody incubation: Incubate the sample (such as a section or cell) with the primary antibody (specific antibody), usually overnight at 4°C, followed by washing with PBS.
2. HRP-labeled secondary antibody incubation: Add the secondary antibody (secondary antibody conjugated to HRP) to the sample and incubate for 30 minutes to 1 hour.
3. Color development: Mix TMB substrate solution with hydrogen peroxide solution and add to the sample. The color development reaction generally lasts 10-30 minutes, depending on the expression level of the target protein and the experimental requirements.
4. Stop reaction: When the color of the blue or green product reaches the desired intensity, use a stop solution (e.g., 0.5 M sulfuric acid) to stop the reaction.
5. Microscope observation: Observe the sample under a microscope, and the target area should show blue or green staining.
III. Application of enzyme-linked immunosorbent assay (ELISA)
1. Blocking: Block the microplate with an appropriate blocking solution (e.g., 5% BSA or bovine serum albumin, BSA) to avoid nonspecific binding.
2. Antibody incubation: Incubate the sample (e.g., serum) with the primary antibody in the microplate coated with the antigen for 1 hour.
3. HRP-labeled secondary antibody incubation: Incubate the sample with a secondary antibody conjugated to HRP, usually for 30 minutes to 1 hour.
4. Color development reaction: Add the pre-prepared TMB substrate solution (a mixture of TMB and hydrogen peroxide) and allow the reaction to occur. The reaction usually lasts for 5-15 minutes until a blue or green product is formed.
5. Stop the reaction: Add a stop solution (e.g. 2 M sulfuric acid) to stop the reaction, and the blue reaction solution will turn yellow.
6. Absorbance measurement: Use a spectrophotometer to measure the absorbance value at a wavelength of 450 nm, which is proportional to the antibody concentration.
Notes:
1. The time and temperature of the color development reaction need to be optimized according to the sample and experimental conditions. It is usually recommended to incubate at room temperature for 10-30 minutes, but in some cases, the reaction time can be appropriately extended or shortened according to actual needs.
2. Too long a reaction time may result in background staining or too strong a color, so it is necessary to monitor and ensure that the reaction is stopped within the optimal reaction time.
The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |

 United States
United States