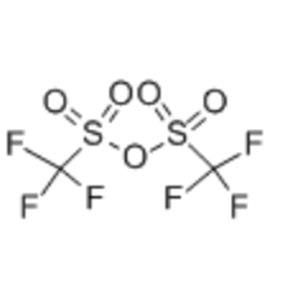
Product Name: Trifluoromethanesulfonic Anhydride (Triflic Anhydride)
CAS No.: 358-23-6
Molecular Formula: C₂F₆O₅S₂ / (CF₃SO₂)₂O
Molecular Weight: 282.13 g/mol
Overview:
Trifluoromethanesulfonic anhydride is the anhydride of triflic acid and an exceptionally reactive, versatile super-acylating and activating agent. Its reactivity far exceeds that of common anhydrides (e.g., acetic anhydride), making it one of the most direct and efficient tools for introducing the triflyl group (-OTf, triflate). The triflate group is among the best leaving groups known, serving as a crucial intermediate for constructing C-C and C-heteroatom bonds. It is renowned in advanced synthesis as the "universal activator".
Key Advantages:
High Purity & Stable Supply: Advanced purification techniques ensure consistent product purity ≥98%. Although highly reactive, it maintains stable shelf life under strict anhydrous, sealed conditions.
Exceptional Reactivity & Performance: As a super-activator, it facilitates highly efficient reactions under relatively mild conditions, significantly improving yield and purity while shortening synthetic routes.
Scalable & Safe Production: With dedicated production lines and stringent safety protocols, we ensure stable and safe supply from gram to kilogram scale.
Competitive Cost & Service Advantage: Compared to imported high-end reagents, our localized production and supply chain offer more competitive pricing and responsive technical support.
Physicochemical Properties:
Appearance: Colorless to pale yellow liquid, fuming vigorously in moist air and hydrolyzing.
Boiling Point: 81-84 °C.
Density: ~1.677 g/cm³ (25°C).
Solubility: Soluble in most organic solvents (e.g., dichloromethane, acetonitrile, THF).
Hazards: Highly corrosive. Reacts violently with water to form corrosive triflic acid. Extremely moisture-sensitive. Must be handled under strict anhydrous conditions with full protective equipment.
Primary Applications:
Preparation of Highly Reactive Intermediates:
Triflates (R-OTf): Converts alcohols, phenols, or enols into corresponding triflates, which serve as superior aryl or vinyl substrates in palladium-catalyzed cross-coupling reactions (e.g., Suzuki, Negishi, Stille), with reactivity far exceeding traditional halides.
N-Triflyl Compounds: Reacts with amines to form highly reactive amide derivatives or for amine protection.
As a Potent Dehydrating/Activating Agent:
Carboxylic Acid Activation: Converts carboxylic acids in situ into mixed anhydrides or more reactive acylimidazolium salts for peptide coupling or synthesis of sterically hindered esters.
Amide/Sulfonamide Activation: Facilitates substitution or cyclization reactions.
Heterocycle Synthesis: Promotes lactonization, lactamization, and the synthesis of various heterocyclic compounds.
As a Triflyl (-SO₂CF₃) Source: For introducing fluorine-containing functional groups into molecules.
Packaging & Storage:
Packaging: Available in a range of sizes (1g, 5g, 10g, 50g, 100g, etc.) in inert gas-filled, sealed bottles with PTFE/silicone septa. Custom larger packaging is available.
Storage: Must be stored under anhydrous, inert atmosphere. Recommend storage at 0-4°C in a sealed container under an inert gas (argon or nitrogen) in a dry, refrigerated environment. For best results, use immediately after opening or re-seal meticulously under controlled conditions.
We welcome inquiries, collaboration, and sample requests from researchers and industrial partners in pharmaceuticals, new materials synthesis, organic methodology, and high-end fine chemicals!
Yurui(Shanghai) Chemical Co. Ltd / UIV Chem
Contact Person: Nan Haodong
Address: Floor 5, Building H2, No.3188 Xiupu Rd, Pudong, Shanghai 201203, China
Tel: 0086-21-50456736 / 021-33191321
WhatsApp: +86 17613296318
WeChat: NHDong2021
Email: xin@riyngroup.com
QQ: 2551819264
Website: www.riyngroup.com

 China
China