Valsartan Impurity
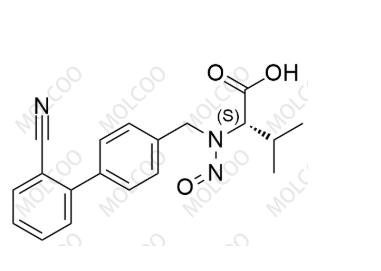
Product Code:V002064
English Name:Valsartan Impurity 64
English Alias:(S)-2-(((2'-cyano-[1,1'-biphenyl]-4-yl)methyl)(nitroso)amino)-3-methylbutanoic acid
CAS No.:2407652-86-0
Molecular Formula:C19H19N3O3
Molecular Weight:337.37
Ultra-High Purity:Confirmed by HPLC (≥99.5%), along with NMR (1H, 13C), HRMS, and X-ray single-crystal diffraction, ensuring accurate and reliable impurity analysis.
Strong Stability:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.1% in acetonitrile - water solution within 6 months, providing high reproducibility of experimental data.
Quality Control Testing:Used for UPLC - MS/MS detection of Impurity 64 in Valsartan API and formulations, strictly controlling impurity content to meet ICH Q3A and Q3B standards (single impurity limit ≤0.1%).
Process Optimization Research:Monitor the formation pathway of Impurity 64 during Valsartan synthesis. Reduce impurity generation by over 60% by adjusting the condensation reaction temperature (e.g., 50 - 60℃), reaction time, and catalyst dosage.
Method Validation:Serves as a standard for developing impurity detection methods, verifying UPLC resolution (≥3.5) and LOD (0.003 ng/mL) to ensure compliance with regulatory requirements.
Valsartan, an angiotensin II receptor antagonist, is widely used in the treatment of hypertension and heart failure. Impurity 64, as a specific process - related impurity of Valsartan, may arise from incomplete intermediate reactions, side reactions, or raw material residues during multi-step synthesis. Its nitroso and cyano groups may affect the drug's stability, safety, and efficacy. After the Valsartan recall incident in 2018, global regulatory agencies (such as FDA and EMA) have imposed stricter requirements on Valsartan impurity control. The study of Impurity 64 has become crucial for ensuring drug quality and patient safety.
Detection Technology:UPLC - MS/MS with a C18 column (1.7μm) and 0.1% formic acid - acetonitrile gradient elution achieves separation within 5 minutes, with an LOD as low as 0.001 ng/mL, enabling highly sensitive detection of trace impurities.
Formation Mechanism:Studies indicate that Impurity 64 mainly forms during the reaction of the cyano-biphenyl intermediate with nitrosating agents. Optimizing the reaction solvent system (e.g., using DMF instead of methanol) and strictly controlling the dosage of nitrosating agents can effectively suppress its formation.
Safety Evaluation:In vitro cytotoxicity tests show that the IC₅₀ of Impurity 64 against HUVEC cells is 125.6 μM (Valsartan IC₅₀ = 8.3 μM). Although less toxic than the main drug, its content in drugs still requires strict control. Long-term stability testing is currently underway to systematically monitor its degradation behavior under different humidity, light, and temperature conditions.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!