Vitamin K1 Impurity 85761-30-4
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
Product Information
Product Number: V017023
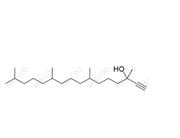
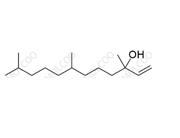
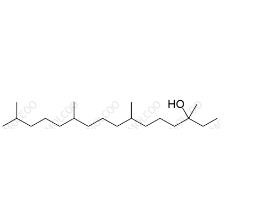
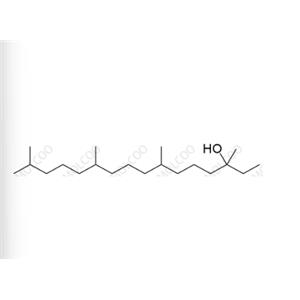
English Name: Vitamin K1 Impurity 23
English Alias: 3,7,11,15-tetramethylhexadecan-3-ol
CAS Number: 85761-30-4
Molecular Formula: C20H42O
Molecular Weight: 298.55
Advantages: As a reference standard for Vitamin K1 Impurity 23, it has an accurate and clear chemical structure, and has undergone strict purity testing and quality control with strong stability, which can maintain stable properties under various storage conditions. It can provide a reliable reference for the quality inspection of Vitamin K1 and its preparations, ensuring the accuracy and repeatability of impurity detection results, effectively helping pharmaceutical companies and research institutions to precisely control drug quality and meet the strict quality requirements of drug research and development and production.
Applications: It is mainly used in the quality research, impurity analysis, and quality control of Vitamin K1 bulk drugs and formulations. It is used to establish and validate impurity detection methods such as high-performance liquid chromatography (HPLC) and gas chromatography - mass spectrometry (GC - MS). During the research and development of Vitamin K1, it helps to study the source and formation mechanism of impurities, and optimize the synthesis process to reduce impurity generation. In the production process, it is used to monitor the content of Impurity 23 in products in real time to ensure that drug quality meets relevant standards and regulatory requirements. It can also be used to evaluate the changes of impurities in Vitamin K1 drugs during storage and transportation, providing data support for drug stability research.
Background Description: Vitamin K1 plays an important role in physiological processes such as blood coagulation and bone health and is a commonly used clinical drug. In the process of its production and research and development, the presence of impurities may affect the safety, effectiveness, and stability of the drug. To ensure the safety of patients' medication and meet the requirements of drug regulation, strict research and precise control of impurities in Vitamin K1 are essential. As one of the impurities of Vitamin K1, in-depth research on Vitamin K1 Impurity 23 helps to improve the quality standard system of Vitamin K1 and enhance drug quality.
Research Status: Currently, the research on Vitamin K1 Impurity 23 mainly focuses on the optimization and improvement of impurity analysis methods. By using more advanced detection technologies and instruments, the sensitivity and accuracy of detecting this impurity are improved. At the same time, researchers are actively exploring the source and change rules of this impurity during the synthesis and storage of Vitamin K1, and reducing the generation and accumulation of impurities by improving process conditions and optimizing the storage environment. In addition, the research on the potential impact of this impurity on the performance and safety of Vitamin K1 drugs is also gradually underway, aiming to provide a more comprehensive scientific basis for the overall evaluation of Vitamin K1 quality.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China