Xanomeline Impurity 5
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Number: X002005
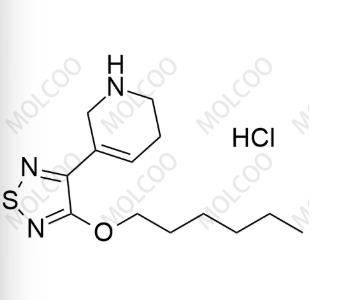
English Name: Xanomeline Impurity 5
English Alias: 3-(hexyloxy)-4-(1-nitroso-1,2,5,6-tetrahydropyridin-3-yl)-1,2,5-thiadiazole
CAS Number: None
Molecular Formula: C₁₃H₂₀N₄O₂S
Molecular Weight: 296.39
As Impurity 5 of Xanomeline, this compound has the following advantages:
Well-defined with distinct polyheterocyclic features: Contains 1,2,5-thiadiazole ring (3-hexyloxy, 4-tetrahydropyridinyl substituted) and 1-nitroso-1,2,5,6-tetrahydropyridine ring. Unlike xanomeline (muscarinic acetylcholine receptor agonist with non-nitroso tetrahydropyridine), its nitroso polarity, thiadiazole sulfur properties, and hexyloxy hydrophobicity create significant differences, enabling precise differentiation via HPLC/GC as a specific marker;
High stability and traceability: Rigid thiadiazole/tetrahydropyridine structures and stability of ether bonds ensure stability under dark, low-temperature conditions. As a byproduct from tetrahydropyridine ring nitrosation during storage/degradation of xanomeline, it directly reflects tertiary amine oxidation activity and nitrite exposure risk, improving process tracing accuracy;
High detection sensitivity: Thiadiazole-tetrahydropyridine conjugation shows strong UV absorption (240-270nm), combined with m/z 297 [M+H]⁺ enabling ppb-level analysis via LC-MS, compatible with muscarinic agonist nitroso impurity systems.
Pharmaceutical quality control: Used as an impurity reference standard to quantify Xanomeline Impurity 5 in APIs, ensuring compliance with limits for potentially harmful impurities;
Stability studies: Monitoring impurity levels under varying conditions (pH, light) to assess nitrosation trends and support safety assurance over shelf life;
Synthesis assessment: Evaluating purity of tetrahydropyridine-containing intermediates in xanomeline synthesis to reduce nitrosation risk at the source.
Xanomeline is a muscarinic acetylcholine receptor agonist containing 1,2,5-thiadiazole and tetrahydropyridine rings. Its tetrahydropyridine tertiary amine (-N-) may undergo nitrosation upon exposure to nitrous acid (e.g., from nitrate conversion) during production or storage, forming 1-nitroso-1,2,5,6-tetrahydropyridine derivatives like Xanomeline Impurity 5. Due to potential toxicity of nitroso compounds, their residues impact xanomeline safety, requiring strict detection and control.
Current research focuses on:
Analytical method validation: Developing HPLC assays with C18 columns for separation, achieving 0.1 ppb detection limits;
Nitrosation mechanism: Studying impurity formation kinetics under varying nitrite concentration and pH to clarify tetrahydropyridine tertiary amine-to-nitroso conversion pathways;
Control strategies: Exploring nitrosation inhibitors (e.g., vitamin C) to keep impurity levels below safety limits;
Toxicity evaluation: Conducting in vitro cell studies to assess potential toxicity and support limit setting
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com