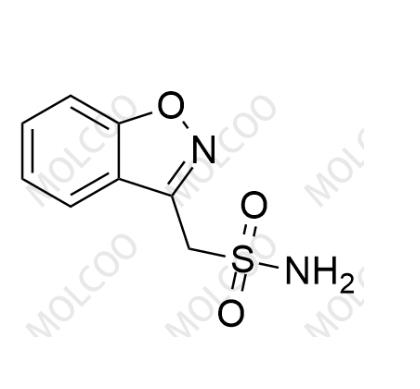
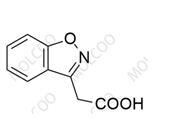
Detection Technology:
The mainstream method is HPLC-UV using a C18 column (4.6×250mm, 5μm) with acetonitrile-phosphate buffer (30:70, v/v) as the mobile phase, detection at 225nm, and a limit of quantification (LOQ) of 0.05%. Developed UPLC-MS/MS technology shortens analysis time to <5 minutes and enables trace impurity detection (e.g., nitrosamines, LOQ as low as 0.01ppm).
Process Optimization:
In traditional processes, the sulfonyl chloride intermediate is prone to hydrolysis to form Impurity 5 (2-(benzo[d]isoxazol-3-yl)acetic acid). Using triethylamine as a base and controlling the reaction temperature ≤5°C reduces this impurity from 0.8% to 0.03%. New catalytic synthesis routes (e.g., palladium-catalyzed coupling) reduce side reactions and increase API purity to >99.8%.
Safety Studies:
Long-term toxicity tests show the no-observed-adverse-effect level (NOAEL) of zonisamide in rats is 200mg/kg/day. Nitroso compounds in the impurity profile must comply with ICH M7(R1) for genotoxic impurities, driving the development of highly sensitive detection methods.





 China
China