
18YR企业会员
发布人:北京百灵威科技有限公司
发布日期:2026/4/13 16:50:55
在药物、农药及功能材料分子中引入二氟甲基(CF2H),可显著提升分子的代谢稳定性、脂溶性及生物利用度,是含氟分子设计与优化的关键策略之一。因此,高效、可控地实现二氟甲基化反应,成为有机氟化学与药物化学领域的核心研究课题。
然而,现有试剂在实际研发中长期存在稳定性差、模式单一、条件苛刻等问题,导致反应产率低、重复性差、适用范围窄,成为含氟分子研发的关键瓶颈。
深圳大学刘国凯副教授团队设计开发的亲电型二氟甲基锍盐试剂——S-(二氟甲基)-苯基-(2,4,6-三甲氧基苯基)硫鎓四氟硼酸盐,通过分子内氢键设计策略,从根本上解决了传统试剂稳定性差的问题,并在多个维度实现全面突破。
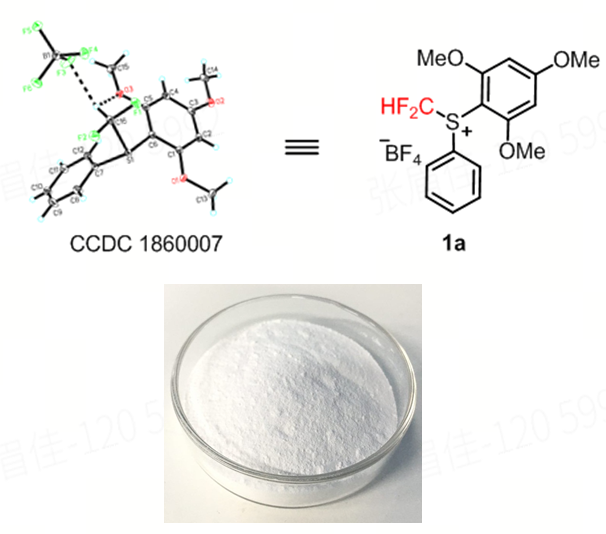
●超高稳定性:结晶性粉末,对空气/湿气不敏感。
●多模式反应:可实现•CF₂H、+CF₂H、:CF₂H三种活性物种的转化,覆盖自由基、亲电、卡宾等多种反应机制。
●底物适用范围广:适用于β-酮酯、醇、酚、异氰、烯烃等多种底物类型,满足不同研发场景需求。
●操作条件温和:常规实验室环境即可使用,适合高通量筛选和规模化应用
●平台型试剂定位:为二氟甲基化新策略、新方法开发提供高效实用的通用工具。
见图2。
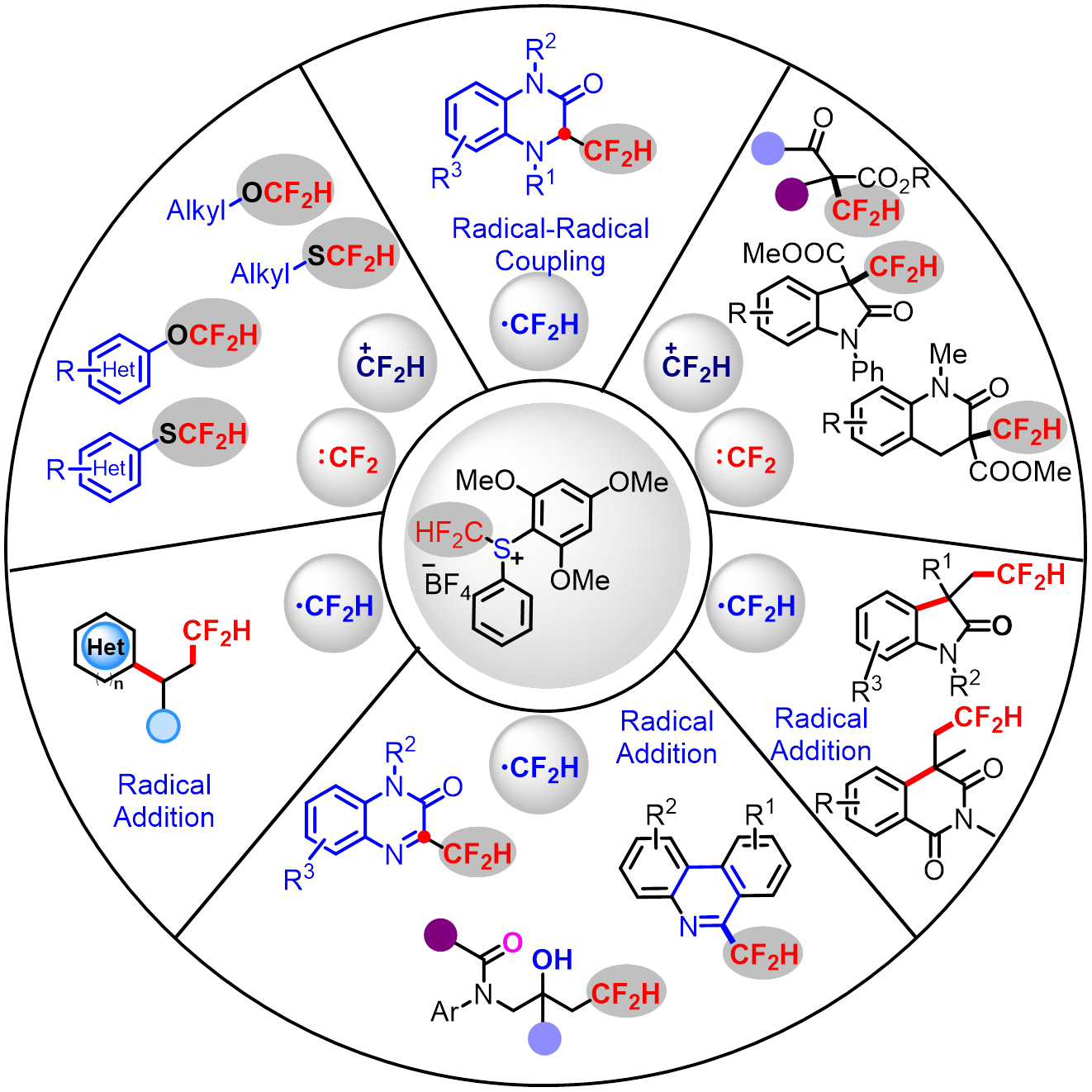

Air- and Light-Stable S-(Difluoromethyl)sulfonium Salts: C-Selective Electrophilic Difluoromethylation of β-Ketoesters and Malonates, Org. Lett. 2018, 20, 6925-6929.
Direct C(sp³)–H difluoromethylation via radical–radical cross-coupling by visible-light photoredox catalysis, Org. Chem. Front., 2022, 9, 2141−2148.
Difluoromethyl Radical Triggered Tandem Reaction of N‑Allyl Amides to Difluoromethylated β‑Amino Alcohols by Photoredox Catalysis, Org. Lett. 2021, 23(21), 8482−8487.
Facile difluoromethylation of aliphatic alcohols with an S-(difluoromethyl)sulfonium salt: reaction, scope and mechanistic study, Chem. Commun. 2019, 55, 7446-7449.
Selective O-difluoromethylation of 1,3-diones using S-(difluoromethyl)sulfonium salt, Chin. Chem. Lett. 2019, 30,1515-1518.
Visible-Light Photoredox-Catalyzed Three-Component Reaction of Carbazoles with Alkenes and S‑(Difluoromethyl)sulfonium Salt: A Practical Approach to Access Difluoroalkyl-Functionalized Carbazoles, J. Org. Chem. 2023, 88, 10711−10720.
Facile Entry to Pharmaceutically Important 3-Difluoromethyl-Quinoxalin-2-Ones Enabled by Visible-Light-Driven Difluoromethylation of Quinoxalin-2-Ones, Pharmaceuticals 2022, 15, 1552.
Visible-Light-Driven Difluoromethylation of Isocyanides with S‑(Difluoromethyl)diarylsulfonium Salt: Access to a Wide Variety of Difluoromethylated Phenanthridines and Isoquinolines, J. Org. Chem. 2020, 85, 10479-10487.
Difluoromethylation of Phenols and Thiophenols with the S-(Difluoromethyl)sulfonium Salt: Reaction, Scope, and Mechanistic Study, J. Org. Chem. 2019, 84, 15948-15957.
相关新闻资讯