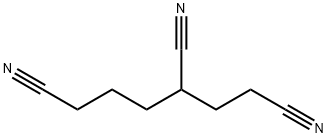
1,3,6-Hexanetricarbonitrile: Electrolyte Additive for Advanced Batteries
1,3,6-hexanetricarbonitrile is a multifaceted chemical compound with critical applications in advanced battery technology, organic synthesis, and polymer science. Its unique properties make it an indispensable component for driving innovation across multiple industries.

An efficient electrolyte additive of 1,3,6-hexanetricarbonitrile
The development of advanced energy storage systems is critically important for the large-scale applications of renewable energy resources with intermittent and volatile power natures (e.g., solar and wind). Nitrile organic compounds containing polar groups as electrolyte additives not only show great promising ion storage properties for ZIBs but also lithium-ion batteries (e.g., succinonitrile (SN), adiponitrile (ADN) and 1,3,6-Hexanetricarbonitrile (HTCN). HTCN is an environmentally friendly polar non-protonic solvent, which is soluble in water, with low viscosity, high boiling point, wide electrochemical window and high chemical stability. In addition, the decomposition products of HTCN in solvent are generally carboxylates, aldehydes and organic amines rather than CN−. As a result, the HTCN has been widely used as an important intermediate for industrial organic synthesis and electrolyte additives for lithium-ion batteries. Herein, we for the first time employed a series of 1,3,6-hexanetricarbonitrile (HTCN-x, x = 0, 0.1, 0.3 and 0.5) with different x volume concentrations as electrolyte additives into zinc trifluoromethanesulphonate electrolytes (Zn(CF3SO3)2 or Zn(OTf)2) and studied their electrochemical properties in ZIB cells with the use of Zn metal as the anode and ZnxV2O5·nH2O (ZVO) as the cathode. The strong interaction between H2O and HTCN could significantly reduce the H2O population of the solvation sheath, leading to reduced side reactions in the aqueous Zn(OTf)2 electrolyte.[1]
In summary, we for the first time prepared a small volume concentration (x) of 1,3,6-hexanetricarbonitrile (HTCN-x, x = 0, 0.1, 0.3 and 0.5) as additives into Zn(OTf)2 electrolyte and studied their electrochemical properties in ZIB cells using Zn as the anode and ZVO as the cathode. Importantly, the electrochemical properties of the cells can be significantly enhanced when the amount of the additive is as low as x = 0.3. The Zn||Zn cell using HTCN-0.3 electrolyte presents excellent cycling stability over 950 h at 1 mA cm−2, 1 mAh cm−2. The electrochemical properties are highly comparable to previous reports on the organic compounds as electrolyte additives for ZIBs. The outstanding properties are attributed to the strong interaction between H2O and 1,3,6-hexanetricarbonitrile that could significantly reduce the population of solvated H2O of the Zn2+ solvation sheath, leading to the reduced side reactions in the aqueous Zn(OTf)2 electrolyte. Moreover, the HTCN additive also facilitates the formation of a strong and stable SEI layer on the surface of Zn metal, which effectively prevents the growth of Zn dendrites and the anode corrosion from the electrolyte. As expected, this work not only demonstrates the feasibility of 1,3,6-hexanetricarbonitrile as an electrolyte additive to significantly enhance the rate capability and cycling life of ZIBs, but also provides guidance for other nitrile organic compounds for aqueous ZIBs in the future.
1,3,6-Hexanetricarbonitrile inhibits sodium dendrites in sodium-metal batteries
Rechargeable sodium-metal batteries (SMBs) have attracted much attention recently because of their low cost, high capacity, and abundant sodium resources. In addition, the solvated shell binding of Na+ is relatively weak, reducing the Na+ desolvation barrier and enhancing the kinetic properties of SMBs. Nevertheless, the inherent challenge of unstable Na+ deposition often results in the overgrowth of sodium dendrites, presenting significant stability and safety concerns for SMBs. Herein, we established a stable SEI layer by incorporating a 3 % 1,3,6-hexanetricarbonitrile electrolyte additive into a 1 M NaPF6 carbonate electrolyte. The SEI layer was enriched with Na3N and other nitrides formed by in-situ reduction of HTCN during cell cycling characterized by X-ray photoelectron spectroscopy (XPS) and scanning electron microscope (SEM). Observations from in-situ optical microscopy revealed the growth of sodium dendrites, with the addition of HTCN effectively inhibiting their disordered development. Furthermore, the electrochemical properties exhibited significant improvement with the incorporation of HTCN. The achieved stability was evident in a Na/Na symmetric cell cycling at a current density of 0.5 mA cm−2. This stability was further demonstrated in a full cell assembled with Na3V2(PO4)3 (NVP)/Na, showcasing both stable cycling performance and high multiplicity performance. The introduction of 1,3,6-hexanetricarbonitrile additives emerges as a promising strategy to achieve highly stable SMBs.[2]
In our experiments, a 1 M NaPF6 electrolyte in a solvent mixture comprising ethylene carbonate (EC), diethyl carbonate (DEC), and fluoroethylene carbonate (FEC) in a ratio of 1:1:0.05 vol% was utilized (EDF). An extra 3 vol% of HTCN was also incorporated for the EDF condition (EDF/HTCN). To evaluate the effectiveness of HTCN on SMBs, we tested different concentrations of 1,3,6-hexanetricarbonitrile on EDF electrolyte ionic conductivity. The conductivity profile exhibited a parabolic trend with increasing HTCN concentration. The addition of HTCN enhances the ionic conductivity and electrochemical stability window of the electrolyte. In addition, it was found by X-ray photoelectron spectroscopy (XPS) and scanning electron microscopy (SEM) that HTCN accelerated Na+ shuttling and inhibited the growth of sodium dendrites by reacting with Na metal to form a SEI layer rich in Na3N and NxOy−. By in-situ optical microscopies, it was observed that the addition of 1,3,6-hexanetricarbonitrile additive could effectively inhibit the disordered growth of sodium dendrites. On the other hand, the addition of HTCN changed the solvation structure in the original electrolyte system and promoted the preferential reaction of 1,3,6-hexanetricarbonitrile with Na metal to generate a stable SEI layer. Electrochemical performance shows excellent cycling and rate performance in a full cell assembled with NVP/Na, with cycle stability (4500 cycles), averaging 99.8 % CE and 92.7 % capacity retention. HTCN additives make it possible to achieve highly stable SMBs.
Nonsacrificial Nitrile Additive for Armoring High-Voltage Cathode
With the rapid development of electric vehicles (EVs), electronic products and other energy storage systems, the demand for high energy density lithium-ion batteries (LIBs) has increased significantly. As reported by Zhao et al., they demonstrated that 1,3,6-hexanetricarbonitrile (HTCN) and tris(trimethylsilyl) phosphate (TMSP) electrolyte additives could significantly improve the overall electrochemical performance of the Li1.13Mn0.517Ni0.256Co0.097O2 cell at high cutoff voltage with the protection of CEI. Wang et al. elaborated the interfacial interaction between HTCN additive and Li1.2Ni0.13Co0.13Mn0.54O2 cathode at high operating voltage by theoretical calculation and ex situ physical characterizations. However, at high voltage such as 4.5 V (vs Li+/Li), the oxidation decomposition of some unstable nitrile additives will influence the CEI layer between nitrile additive and Ni-rich cathodes. Herein, two kinds of nitrile compounds, 1,3,6-hexanetricarbonitrile and 1,2-Bis(2-cyanoethoxy)ethane (DENE), are utilized as model electrolyte additives for studying the relationships between TM coordination effect and additive-derived CEI layer. HTCN shows high antioxidation and strong interactions with TM atoms, thus participating in the formation of reliable CEI layer, whereas DENE will preferentially decompose and cause continuous electrolyte side reactions possibly due to the presence of protic species.[3]
In conclusion, this work demonstrates two different nitrile additives of HTCN and DENE in the commercial electrolyte to modify NCM83 cathode and exhibits the enhanced electrochemical performances. 1,3,6-hexanetricarbonitrile has high antioxidation ability, whereas DENE is preferentially oxidized. The results of electrochemical performance of the NCM83 cathode with BE-HTCN electrolyte have been greatly enhanced compared with the BE and BE-DENE electrolytes. This improvement is mainly attributed to the formation of the stable and effective electrode/electrolyte interface on the NCM83 cathode by the introduction of 1,3,6-hexanetricarbonitrile additive, which can anchor on the NCM83 surface as a CEI framework, suppress the decomposition of the electrolyte and enhance the structural stability of the cathode material.
References
[1]Wang, Rui et al. “An efficient electrolyte additive of 1,3,6-hexanetricarbonitrile for high performance aqueous zinc-ion batteries.” Journal of colloid and interface science vol. 646 (2023): 950-958. doi:10.1016/j.jcis.2023.05.072
[2]You, J., Xu, Z., Zhang, Y., Li, Y., Zhang, B., Cao, Y., Deng, L., Li, T., & Wang, W. (2024). 1,3,6-Hexanetricarbonitrile as electrolyte additive to inhibit sodium dendrites in sodium-metal batteries. Chemical Engineering Journal, 492, 152198. https://doi.org/10.1016/j.cej.2024.152198
[3]Li, Xin et al. “Nonsacrificial Nitrile Additive for Armoring High-Voltage LiNi0.83 Co0.07 Mn0.1 O2 Cathode with Reliable Electrode-Electrolyte Interface toward Durable Battery.” Small (Weinheim an der Bergstrasse, Germany) vol. 18,30 (2022): e2202989. doi:10.1002/smll.202202989
You may like
See also
Lastest Price from 1,3,6-HEXANETRICARBONITRILE manufacturers
US $1.10-9.90/kg2025-08-18
- CAS:
- 1772-25-4
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 100kg

US $20.00-8.00/kg2025-03-07
- CAS:
- 1772-25-4
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 5 tons