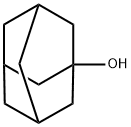
1-Adamantanol: Inclusion Complexation & Antimycobacterial Applications
1-Adamantanol is a cyclic molecule with a hydroxyl group that can be used as an intermediate in the synthesis of adamantane. 1-Adamantanol can be oxidized to 1,3-adamantanediol by the Streptomyces SA8 Oxidation System.

1-Adamantanol as Molecular knots
The formation of inclusion complexes of 6-(N-methyl-3-phenylpropionamido)-βCD and 6-(N-methylcinnamido)-βCD with 1-adamantanol was investigated in detail by 1H NMR.213 In the case of the 6-(N-methyl-3-phenylpropionamido)-βCD, the amide linkage preferably took the Z-form to form the stable self-inclusion complex. In the presence of 1-adamantanol, the βCD derivative formed a stable inclusion complex with 1-adamantanol, leading to a reduced relative stability of the amide Z-form. These observations indicate that 1-adamantanol regulated the balance of amide Z- and E-forms. In the case of the 6-(N-methylcinnamido)-βCD, on the other hand, trans-to-cis and cis-to-trans photoisomerization of the cinnamoyl moiety switched the balance of amide Z- and E-forms. The amide linkage in the cinnamoyl cis isomer favored adoption of the Z-form to form a very stable self-inclusion complex.[1]
In the presence of 1-adamantanol, the formation of complexes with 1-adamantanol reduced the relative stability of the amide Z-form. In the case of the cinnamoyl trans isomer, however, 1-adamantanol did not regulate the balance of the amide Z- and E-forms significantly. βCD derivatives modified with a PEG-carrying cinnamoyl moiety at the 6-position through an ester linkage were synthesized. The βCD derivatives formed stable self-inclusion complexes, in which the cinnamoyl moiety was included in the βCD cavity. In the presence of an equivalent of 1-adamantane carboxylic acid (AdCA), βCD derivatives formed stable inclusion complexes with AdCA. It is noteworthy that, in the presence of a half equivalent of AdCA, the exchange between the self-inclusion complex and inclusion complex with AdCA was observed on the NMR time scale. The exchange rate constants were evaluated by 1H NMR at different temperatures and two-dimensional exchange spectroscopy to study the kinetics of self-threading. The exchange rate constant decreased exponentially with increasing the degree of polymerization of PEG.
Antimycobacterial Evaluation of Novel 1-Adamantanol Analogues
The treacherous nature of tuberculosis (TB) combined with the ubiquitous presence of the drug-resistant (DR) forms pose this disease as a growing public health menace. Therefore, it is imperative to develop new chemotherapeutic agents with a novel mechanism of action to circumvent the cross-resistance problems. The unique architecture of the Mycobacterium tuberculosis (M. tb) outer envelope plays a predominant role in its pathogenesis, contributing to its intrinsic resistance against available therapeutic agents. The mycobacterial membrane protein large 3 (MmpL3), which is a key player in forging the M. tb rigid cell wall, represents an emerging target for TB drug development. Several indole-2-carboxamides were previously identified in our group as potent anti-TB agents that act as inhibitor of MmpL3 transporter protein. Despite their highly potent in vitro activities, the lingering Achilles heel of these indoleamides can be ascribed to their high lipophilicity as well as low water solubility. In this study, we report our attempt to improve the aqueous solubility of these indole-2-carboxamides while maintaining an adequate lipophilicity to allow effective M. tb cell wall penetration. A more polar 1-Adamantanol moiety was incorporated into the framework of several indole-2-carboxamides, whereupon the corresponding analogues were tested for their anti-TB activity against drug-sensitive (DS) M. tb H37Rv strain. Three 1-Adamantanol derivatives showed nearly 2- and 4-fold higher activity (MIC = 1.32 – 2.89 μM) than ethambutol (MIC = 4.89 μM).[2]
Remarkably, the most potent 1-Adamantanol analogue demonstrated high selectivity towards DS and DR M. tb strains over mammalian cells [IC50 (Vero cells) ≥ 169 μM], evincing its lack of cytotoxicity. The top eight active compounds retained their in vitro potency against DR M. tb strains and were docked into the MmpL3 active site. The most potent 1-Adamantanol/adamantane-based indoleamides displayed a two-fold surge in potency against extensively DR (XDR) M. tb strains with MIC values of 0.66 and 0.012 μM, respectively. The adamantanol-containing indole-2-carboxamides exhibited improved water solubility both in silico and experimentally, relative to the adamantane counterparts. Overall, the observed antimycobacterial and physicochemical profiles support the notion that 1-Adamantanol moiety is a suitable replacement to the adamantane scaffold within the series of indole-2-carboxamide-based MmpL3 inhibitors.
References
[1]Hashidzume, A., Tokashima, Y., Yamaguchi, H., & Harada, A. (2017). 1.12 - Cyclodextrin. In Comprehensive Supramolecular Chemistry II (pp. 269-316). Elsevier. https://doi.org/10.1016/B978-0-12-409547-2.13829-6
[2]Alsayed SSR, Lun S, Payne A, Bishai WR, Gunosewoyo H. Design, synthesis and antimycobacterial evaluation of novel adamantane and adamantanol analogues effective against drug-resistant tuberculosis. Bioorg Chem. 2021 Jan;106:104486. doi: 10.1016/j.bioorg.2020.104486. Epub 2020 Nov 19. PMID: 33276981; PMCID: PMC7775894.
You may like
Lastest Price from 1-Adamantanol manufacturers

US $0.00/kg2025-09-28
- CAS:
- 768-95-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1

US $1.10/g2025-09-17
- CAS:
- 768-95-6
- Min. Order:
- 1g
- Purity:
- 99.9%
- Supply Ability:
- 100 Tons Min