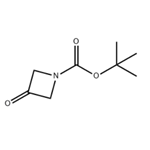
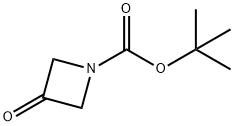
1-Boc-3-azetidinone: Boc-Protected Drug Synthesis Intermediate
1-Boc-3-azetidinone is a key quaternary nitrogen-containing heterocyclic intermediate of great value in the fields of organic synthesis and drug discovery. The core structure of this compound is a nitrogen-containing cyclobutane ring, with an active ketone carbonyl group at the 3-position and the nitrogen atom protected by a tert-butoxycarbonyl (Boc) group. Its unique ring strain structure confers moderate and controllable reactivity, whilst its overall physicochemical properties are stable and compatible with most organic synthesis reaction systems. Industrial and laboratory syntheses typically use 3-azetidinone hydrochloride as the starting material, prepared via a selective protection reaction with Boc anhydride; the process is well-established and product purity is easily controlled. Core value of 1-Boc-3-azetidinone lies in the field of drug synthesis, where the ketone group at the 3-position can undergo various derivatisation reactions such as reduction, nucleophilic addition and amination. It serves as a key building block for constructing JAK inhibitors such as baricitinib, antidepressant 3-aminoazetidine derivatives, anticancer pyruvate kinase M2 inhibitors and antibacterial drugs. 1-Boc-3-azetidinone effectively optimises the metabolic stability and receptor selectivity of drug molecules. During storage, it must be protected under an inert gas atmosphere and kept away from moisture and strongly oxidising environments to prevent group decomposition from affecting reaction efficiency.

Neuroprotective activity of 3-aryl-3-azetidinyl acetic acid methyl ester derivatives
A library of 3-aryl-3-azetidinyl acetic acid methyl ester derivatives was prepared from 1-Boc-3-azetidinone employing the Horner-Wadsworth-Emmons reaction, rhodium(I)-catalyzed conjugate addition of arylboronic acids, and subsequent elaborations to obtain N-unprotected hydrochlorides, N-alkylated and N-acylated azetidine derivatives. The compounds were evaluated for acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) inhibitory activity, revealing several derivatives to possess AChE inhibition comparable to that of the AChE inhibitor rivastigmine. The binding mode of the AChE inhibitor donepezil and selected active compounds within the active site of AChE was studied using molecular docking. Furthermore, the neuroprotective activity of the prepared compounds was evaluated in models associated with Parkinson's disease (salsolinol-induced) and aspects of Alzheimer's disease (glutamate-induced oxidative damage). Benzyl isobutyrate showed the highest neuroprotective effect in both salsolinol- and glutamate-induced neurodegeneration models, and its protective effect in the glutamate model was revealed to be driven by a reduction in oxidative stress and caspase-3/7 activity. A small library of 3-aryl-3-azetidinyl acetic acid methyl ester derivatives was prepared from common starting material 1-Boc-3-azetidinone, employing the Horner-Wadsworth-Emmons reaction, rhodium(I)-catalyzed conjugate addition of arylboronic acids, and subsequent elaborations to obtain N-unprotected hydrochlorides, N-alkylated and N-acylated azetidine derivatives. Compounds were evaluated for AChE and BChE inhibitory activity.[1]
Photochemical Decarbonylation of Oxetanone and Azetidinone
Rich chemistry of 1,3-dipoles such as carbonyl ylides (CY) and azomethine ylides (AMY) rests on reliable methods by which these reactive intermediates can be obtained. The strategy we describe here is based on structural insights obtained from electronic and vibrational spectra, and computational simulations. Like other ketones, UV-vis absorption spectra of 3-oxetanone (3-OX), 1-Boc-3-azetidinone (3-AZ), and cyclobutanone (CB) show a weak absorption centered around 280 nm with extinction coefficients between 10 and 20M−1cm−1. We anticipated that the excitation of the 220 nm-band would lead to chemistry other than decarbonylation, or that it would slow down the desired reaction, so we prevented this excitation by using a filter. Exposing solutions of 3-oxetanone, 1-Boc-3-azetidinone, and cyclobutanone in acetonitrile to ultraviolet light filtered to block wavelengths shorter than 280 nm led to a steady release of carbon monoxide that could be detected within minutes (depending on substrate concentration) in the headspace of the reactor with a commercial electrochemical CO-specific monitor. Photoexcitation of cyclic ketones leads to the expulsion of carbon monoxide and a mixture of products derived from diradical intermediates. Here we show that synthetic utility of this process is improved if strained heterocyclic ketones are used. Photochemistry of 3-oxetanone and 1-Boc-3-azetidinone has not been previously described. Decarbonylation of these 4-membered rings proceeds through a step-wise Norrish type I cleavage of the C–C bond from the singlet excited state. Ylides derived from both compounds are high-energy species that are kinetically stable long enough to undergo [3+2] cycloaddition with a variety of alkenes and produce substituted tetrahydrofurans and pyrrolidines.[2]
References
[1]Šachlevičiūtė, Urtė et al. “Synthesis and neuroprotective activity of 3-aryl-3-azetidinyl acetic acid methyl ester derivatives.” Archiv der Pharmazie vol. 356,12 (2023): e2300378. doi:10.1002/ardp.202300378
[2]Singh M, Dhote P, Johnson DR, Figueroa-Lazú S, Elles CG, Boskovic Z. Photochemical Decarbonylation of Oxetanone and Azetidinone: Spectroscopy, Computational Models, and Synthetic Applications. Angew Chem Int Ed Engl. 2023 Jan 16;62(3):e202215856. doi: 10.1002/anie.202215856. Epub 2022 Dec 13. PMID: 36399366; PMCID: PMC9839639.
You may like
See also
Lastest Price from 1-Boc-3-azetidinone manufacturers

US $0.00-0.00/KG2025-04-21
- CAS:
- 398489-26-4
- Min. Order:
- 1KG
- Purity:
- ≥98.0%
- Supply Ability:
- Ton
US $0.00-0.00/KG2025-04-04
- CAS:
- 398489-26-4
- Min. Order:
- 1KG
- Purity:
- 98%
- Supply Ability:
- 1ton
![41532-84-7 1,1,2-Trimethyl-1H-benz[e]indoleTMBIheterocyclic intermediate](/NewsImg/2026-04-02/6391072477188175017456875.jpg)
