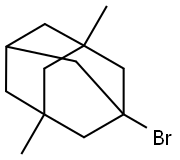
1-Bromo-3,5-dimethyladamantane: Synthesis & Pharmaceutical Applications
1-Bromo-3,5-dimethyladamantane is a typical bridged adamantane bromide derivative featuring a rigid cage structure and high chemical stability, serving as a crucial intermediate in organic synthesis. This compound exhibits excellent reactivity in nucleophilic substitution reactions, playing a pivotal role particularly in the synthetic route of the drug molecule memantine. Consequently, it has garnered significant attention in the fields of fine chemicals and pharmaceutical synthesis.

synthesis of 1,3-dicarbonyl adamantanes from 1-bromoadamantanes
Replacement of labile atoms or groups by other functionalities is a powerful synthetic approach in modern organic chemistry. Meanwhile, selective one-pot bifunctionalization involving the cleavage of both labile C–X and poorly reactive Csp3–H bonds with the formation of compounds containing two new functional groups is a considerable challenge. Principally, depending on the type of the initiating system, these transformations should proceed either through biradical or dication intermediates. We found that carbonylation of 1-bromoadamantane 1a and 1-bromo-3,5-dimethyladamantane 1b in the presence of CBr4·2AlBr3 followed by the in situ treatment of the resulting 1,3-dicarbonylation products with nucleophiles provides a con venient synthetic route to dicarbonyl adamantanes with two new functional groups at the bridged-head position. In contrast to the reactions previously described,3,4 where the original func tionalities interacted with the superelectrophile reversibly and finally remained intact, the here studied labile Br atom was replaced by the carbonyl moiety either. Treatment of 1-bromoadamantane and 1-bromo-3,5-dimethyladamantane with CBr4·2AlBr3 under carbon monoxide atmosphere followed by quenching with nucleophiles affords 1,3-dicarbonyl adamantanes.[1]
1-Bromo-3,5-dimethyladamantane behaved similarly to form the corresponding dicarbonyl compounds. The reactions with 1-Bromo-3,5-dimethyladamantane required somewhat higher excess of E than those with 1a to achieve good selectivity and high yields of the products. It seems likely that the carbonylation product generated from 1-Bromo-3,5-dimethyladamantane is less stable in comparison with that prepared from 1-bromoadamantane, because donor methyl groups in acylium cations facilitate their decarbonylation. The herein developed method seems to be rather common for the preparation of various 1,3-dicarbonyl adamantanes by using nucleophiles which are reluctant to superelectrophiles. In the case of very active nucleophiles, the corresponding compounds can be prepared by the known multi-step reactions starting from adamantanedicarboxylic acids, which could be obtained by our method using water as a nucleophile. In summary, the use of powerful superelectrophilic complex CBr4·2AlBr3 allowed us to achieve selective one-pot bifunctionalization of 1-bromoadamantanes. Thus, a new simple one-pot syn thesis of valuable 1,3-dicarbonyl adamantanes has been developed.
Process for the preparation of 1-bromo-3,5-dimethyladamantane
The present invention relates to an improved process for the preparation of 1-bromo-3,5-dimethyladamantane of formula, which is an useful intermediate for synthesis of 1-amino-3,5-dimethyl adamantane of formula or pharmaceutically acceptable salt thereof. 1-amino-3,5-dimethyl adamantane of formula is commonly known as Memantine. Memantine is adamantane derivative. It is an orally active NMDA (N-methyl D-aspartate) receptor antagonist. Memantine is the first in a novel class of Alzheimer's disease medications acting on the glutamatergic system by blocking NMDA glutamate receptors. It is indicated for the treatment of moderate to severe dementia of the Alzheimer's type. Eli Lilly is innovator of Memantine hydrochloride. It is market in US by Forest Labs under the brand name NAMENDA. The process for preparation of Memantine hydrochloride as disclosed in the product patent is as follows: The reaction involves carrying out bromination of 1,3-dimethyl adamantane, using bromine under reflux conditions to obtain 1-bromo-3,5-dimethyladamantane of formula. Compound of formula is further acylated using acetonitrile and sulphuric acid to obtain N-acetamido-3,5-dimethyl adamantane of formula which is further hydrolyzed and converted to Memantine hydrochloride salt-HCl.[2]
Here's the 1-bromo-3,5-dimethyladamantane preparation details: Charge 1,3-dimethyl adamantane (100 gm. 0.6 moles) at 25-30° C. Charge HBr in AcOH (1 ml) at 25-30° C. Heat reaction mixture up to 50-55° C. Add drop-wise bromine (124.7 ml, 2.41 moles) slowly at 50-55° C. Maintain it for 12 hours at 50-55° C. Distil out excess bromine atmospherically up to 85° C. Cool down the reaction mixture to 25-30° C. Add MDC (800 ml) into it. Stir for 30 minutes at 25-30° C. Cool MDC reaction mixture to 5° C. Added drop wise previously prepared 1500 ml 5% solution of sodium hydrosulfite in DM Water into reaction mixture. Separate the MDC layer and discard aqueous layer. Wash it twice with DM water (100 ml). Distil out MDC completely atmospherically up to 55° C. Remove the traces of MDC under vacuum (50-100 mm) at 50-65° C. Oily residue of 1-bromo-3,5-dimethyladamantane is obtained.
References
[1]Akhrem, I. S., Mikhailov, A. A., & Khusnutdinov, R. I. (2011). The first one-pot synthesis of 1,3-dicarbonyl adamantanes from 1-bromoadamantanes. Mendeleev Communications, 21(5), 259–261.
[2]Suresh, B., & Reddy, P. S. (2011). Process for the preparation of 1-bromo-3,5-dimethyladamantane and memantine hydrochloride (U.S. Patent No. US20110306796A1). United States Patent and Trademark Office.
You may like
Lastest Price from 1-Bromo-3,5-dimethyladamantane manufacturers

US $10.00-8.00/kg2026-01-16
- CAS:
- 941-37-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $0.00-0.00/kg2025-09-26
- CAS:
- 941-37-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1