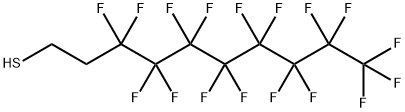
1H,1H,2H,2H-Perfluorodecanethiol: Surface Modification & Functional Applications
1H,1H,2H,2H-Perfluorodecanethiol serves multiple purposes, making it a versatile compound for various applications. When used to modify the surface of polydopamine-silver nanoparticle arrays, it enhances surface-enhanced Raman scattering detection capabilities. This liquid can act as an anti-corrosive coating on copper surfaces, preventing oxidation and providing lubricating properties. This compound forms self-assembled monolayers on gold electrodes, showcasing a high water contact angle and work function, which are beneficial for fabricating organic thin film transistors. Although its anti-corrosion properties may not be as robust as those of n-dodecanethiol-based SAMs due to lower packing density and more defects, it remains effective for intended applications. Furthermore, 1H,1H,2H,2H-perfluorodecanethiol can be grafted onto 1,2-polybutadiene to create transparent antismudge coatings with thermally assisted self-healing capabilities, imbued with a "liquid-like" surface that allows various liquids to effortlessly glide off without leaving residue.

Control of the Redox Activity of Quantum Dots
Modification of the organic adlayer on the surface of a semiconductor nanocrystal (or quantum dot, QD) is a convenient and versatile method for controlling the interactions at the interface of the QD core and its surrounding environment. The specificity of a QD’s interaction with its environment is enhanced if the permeability of the ligand coating is sensitive to both the size and the chemical structure of small-molecule analytes or potential adsorbates. Here, we show that incorporating a partially fluorinated ligand, 1H,1H,2H,2H-perfluorodecanethiol (PFDT) into a small fraction of the organic adlayer of a near-infrared-emitting PbS QD dramatically reduces the efficiency of photo-oxidation of the QD by both 2,3,5,6-tetramethyl-p-benzoquinone (Me4BQ) and 2,6-dimethyl-p-benzoquinone (Me2BQ), due to a decrease in the permeability of the ligand shell to those small molecules. In this work, we quantitatively analyze the permeability of partially fluorinated SAMs on the surfaces of PbS QDs by monitoring the efficiency of collisionally gated photoinduced electron transfer from the QD to an alkyl-substituted benzoquinone (s-BQ). The s-BQ molecule must diffuse through the ligand shell within the lifetime of the PbS exciton in order to extract an electron from the QD core. We find that 1H,1H,2H,2H-Perfluorodecanethiol dramatically reduces this permeability, even when it occupies a small fraction of the ligand shell, due to both its large molecular volume and conformational rigidity and its oleophobicity.[1]
In summary, we used a combination of NMR, photoluminescence, and transient absorption spectroscopies to demonstrate that introducing the fluorinated thiol 1H,1H,2H,2H-Perfluorodecanethiol into the oleate ligand shell on the surfaces of PbS quantum dots is more effective at reducing the permeability of this organic adlayer to solution phase quenchers than is doping the monolayer with the corresponding aliphatic thiol. As probed by the yield of interfacial electron transfer between the QD and a substituted benzoquinone photo-oxidant (Me4BQ or Me2BQ), introducing PFDT into the oleate ligand layer at a surface density of 0.62 PFDT/nm2 (approximately 21% surface coverage) reduces the probability that Me4BQ will permeate the ligand shell (upon collision with its outer surface) by ∼65%, and a surface density of 1.4 PFDT/nm2 (approximately 42% surface coverage) reduces this probability by ∼90%. We determined that the protective properties of the 1H,1H,2H,2H-Perfluorodecanethiol-doped monolayer are due to both the oleophobicity and the steric bulk and rigidity of the PFDT ligand, and that the 1H,1H,2H,2H-Perfluorodecanethiol ligands probably exist in small clusters spread homogeneously over the QD surface. We note that in this analysis, we have not explicitly addressed a theoretical result that indicates that the distance dependence of charge transfer reactions can take different functional forms depending on the driving force for the reaction.
Surface thiolation of silicon for antifouling application
Biofouling is a complex process that involves living organisms and cells probing and attaching to surfaces. Biofouling is a big challenge for the biomedical industry because biofilms form easily on surfaces such as door handles, surgical equipment, and many other medical devices and could increase the spread of disease in humans. Silicon materials are integral parts of our daily lives and have widespread applications in healthcare and manufacturing due to silicon’s unique material properties, including high flexibility, chemical and thermal stability, and ease of fabrication. In addition, silicon materials are mechanically and chemically resilient-able to resist wear in aqueous and organic environments-and display good electrical properties. Therefore, in this study, silicon substrate was selected as a model. Previously, we had developed a technique that allowed us to coat thiol-terminated silicon substrate with 1H,1H,2H,2H-Perfluorodecanethiol molecules through disulfide bonds. Here, the antifouling property of the 1H,1H,2H,2H-Perfluorodecanethiol-coated silicon substrate was tested by aging the substrate in Escherichia coli (E. coli) and Botryococcus braunii (B. braunii) cultures respectively. A large amount of B. braunii colonies were found anchored on the substrate in a 30-day immersion test. However, by applying a reducing agent, the disulfide bonds could be cleaved and the fouled coating could be removed, therefore exposing a non-fouled silicon substrate.[2]
To evaluate the antifouling performance of 1H,1H,2H,2H-Perfluorodecanethiol modified silicon substrate, we immersed the PFDT-coated silicon wafer and Piranha solution (one part 98% H2SO4 and two parts 30% hydrogen peroxide) cleaned silicon wafer in a B. braunii culture. As shown in Fig. 2, after culturing for 1 week, there were large amount of algal cells adhered on the Piranha solution cleaned silicon wafer. In summary, PFDT molecules were integrated onto thiol-terminated silicon substrate through the formation of disulfide bonds. The 1H,1H,2H,2H-Perfluorodecanethiol modified silicon substrate appeared to possess, to some extent, a micro-organism resistant property. However, as the time for the immersion test increased, the overall B. braunii cell density on the PFDT modified silicon substrate increased indicating its antifouling property cannot last forever. It was found that the adhered B. braunii on PFDT modified silicon substrate can be removed by applying TCEP·HCl solution. TCEP·HCl serves as a reducing reagent and can therefore break the disulfide bonds and detach the 1H,1H,2H,2H-Perfluorodecanethiol coating, along with the B. braunii cells adhered on it. This presented approach provides a rational design for removing antifouling coating that becomes aged, all without damaging the original substrate.
Polyurethane surface modified with perfluoroalkyl
Commercially available coatings containing silver nanoparticles embedded in a polymer matrix have been shown to exhibit active antimicrobial behavior in both in vitro and in vivo studies.12 In this study, we aimed to render polyurethane with good antibacterial properties and hemocompatibilties by coating 1H,1H,2H,2H-Perfluorodecanethiol (F-SH) and Ag nanoparticles onto polyurethane films via a reactive polydopamine (PDA) coating.[3]
Inspired by mussel-adhesion phenomena in nature, a simple, mild surface modification process was elaborated to endow the polyurethane (PU) substrate with antibacterial/antifouling properties. In the present study, polydopamine was coated directly onto polyurethane surfaces. AgNO3 was then added and absorbed onto the surface by the active catechol and amine groups of the polydopamine coating. Meanwhile, the adsorbed Ag+ ions were reduced in situ into metallic silver nanoparticles by the "bridge" of the polydopamine coating which yielded a coating with good antimicrobial properties. Finally, 1H,1H,2H,2H-Perfluorodecanethiol (CF3 (CF2 )7 CH2 CH2 SH, F-SH) was attached on the PDA coating via the Michael addition reaction. The hydrophobic F-SH layer above the antibacterial layer yielded a surface with excellent antifouling properties. Preliminary antibacterial assays indicate that the coated surfaces show enhanced antibacterial activity against Escherichia coli (Gram-negative bacteria) and Staphylococcus aureus (Gram-positive bacteria). Platelet adhesion was significantly reduced for the 1H,1H,2H,2H-Perfluorodecanethiol-coated PU film. These results suggest that the modified PU could be used as an antibacterial material for future biomedical applications.
References
[1]Weinberg DJ, He C, Weiss EA. Control of the Redox Activity of Quantum Dots through Introduction of Fluoroalkanethiolates into Their Ligand Shells. J Am Chem Soc. 2016 Feb 24;138(7):2319-26. doi: 10.1021/jacs.5b13077. Epub 2016 Feb 11. PMID: 26820492.
[2]Zhang X, Gao P, Hollimon V, Brodus D, Johnson A, Hu H. Surface thiolation of silicon for antifouling application. Chem Cent J. 2018 Feb 7;12(1):10. doi: 10.1186/s13065-018-0385-6. PMID: 29411153; PMCID: PMC5801134.
[3]Xu D, Su Y, Zhao L, Meng F, Liu C, Guan Y, Zhang J, Luo J. Antibacterial and antifouling properties of a polyurethane surface modified with perfluoroalkyl and silver nanoparticles. J Biomed Mater Res A. 2017 Feb;105(2):531-538. doi: 10.1002/jbm.a.35929. Epub 2016 Nov 3. PMID: 27737518.
You may like
See also
Lastest Price from 1H,1H,2H,2H-Perfluorodecanethiol manufacturers

US $6.00/kg2025-04-21
- CAS:
- 34143-74-3
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2000KG/Month

US $0.00-0.00/KG2025-04-15
- CAS:
- 34143-74-3
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg