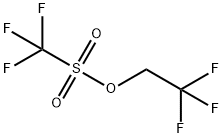
2,2,2-Trifluoroethyl Trifluoromethanesulfonate: Reagent & Electrolyte Component
2,2,2-Trifluoroethyl trifluoromethanesulfonate is a fluorinated organic sulfonate compound which is employed as a trifluoroethylating reagent, it efficiently introduces trifluoroethyl functional groups into molecules. It finds extensive application in pharmaceuticals, pesticides, and functional material synthesis. For instance, in fluorinated drug molecule construction, trifluoroethyl introduction enhances metabolic stability and biological activity.

2,2,2-Trifluoroethyl trifluoromethanesulfonate enables high-voltage Li-ion batteries
Lithium-ion batteries (LIBs) with high energy density (>300 Wh kg−1) and long-term cycling performance are urgently needed for consumer electronics and electric vehicle applications. Extensive researches have been conducted to improve the cathode–electrolyte compatibility. Surface coating or doping the cathodes can reduce the incidence of side reactions between the electrolyte and the cathode. Herein, to address the above challenges, we rationally create an electrolyte, namely 1.9 M lithium bis(fluorosulfonyl)imide (LiFSI) in a mixture of 2,2,2-trifluoroethyl trifluoromethanesulfonate (TTMS) and 2,2,2-trifluoroethyl methanesulfonate (TM) (1:2 v/v). The designed electrolyte forms thin and compact interphases (CEI and SEI) in situ; their high Li+ conductivity ensures fast Li+ intercalation/deintercalation kinetics while the CEI limits the reactivity of the cathode at high potentials. Introducing an electron-withdrawing –CF3 moiety on the β–carbon of ETS endows the resulting 2,2,2-Trifluoroethyl trifluoromethanesulfonate with higher anti-oxidation stability and flame retardancy, and it also reduces the negative effect on the solvation ability of O=S=O groups. To further enhance the solvation of Li-ions, the –CF3 moiety next to the O=S=O is replaced by a –CH3 moiety, thus forming TM. Based on this design rationale, TM should possess high oxidation stability, good Li-ion solvation ability, and the capability to form low-resistance films on both the anode and the cathode simultaneously.[1]
Thanks to the thin and compact SEI, the SEI layer resistance (RSEI) of the cycled graphite anodes retrieved from full cells after 1000 cycles in the 1.9 M LiFSI/2,2,2-trifluoroethyl trifluoromethanesulfonate-2,2,2-trifluoroethyl methanesulfonate electrolyte is only about 6% of that in the 1 M LiPF6/EC–DMC + 2%VC electrolyte. The lower RSEI indicates higher Li+ diffusion kinetics through the SEI. Density functional theory (DFT) calculations were performed to further reveal the underlying mechanism behind the fast kinetics enabled by the 1.9 M LiFSI/TTMS–TM electrolyte. Reduction potential calculations for the explicit presence of Li+ and solvents indicate that LiFSI becomes reductively unstable below 2.84 V (vs. Li+/Li), which is substantially higher than the reduction potential of 2,2,2-Trifluoroethyl trifluoromethanesulfonate (0.31 V vs. Li+/Li) and TM (0.16 V vs. Li+/Li). Thanks to the thin and compact SEI, the SEI layer resistance (RSEI) of the cycled graphite anodes retrieved from full cells after 1000 cycles in the 1.9 M LiFSI/TTMS–TM electrolyte is only about 6% of that in the 1 M LiPF6/EC–DMC + 2%VC electrolyte. The lower RSEI indicates higher Li+ diffusion kinetics through the SEI. Density functional theory (DFT) calculations were performed to further reveal the underlying mechanism behind the fast kinetics enabled by the 1.9 M LiFSI/2,2,2-trifluoroethyl trifluoromethanesulfonate-2,2,2-trifluoroethyl methanesulfonate electrolyte. Reduction potential calculations for the explicit presence of Li+ and solvents indicate that LiFSI becomes reductively unstable below 2.84 V (vs. Li+/Li), which is substantially higher than the reduction potential of TTMS (0.31 V vs. Li+/Li) and TM (0.16 V vs. Li+/Li).
The formulation of electrolytes with superior SEI-forming capability and high oxidative stability can be achieved by choosing solvents in the region where TM and 2,2,2-Trifluoroethyl trifluoromethanesulfonate are situated. This design principle, along with the derived 1.9 M LiFSI/TTMS–TM electrolyte, opens a research avenue for high-voltage and long-cycling LIBs. In summary, by fusing the merits of additive (PS) and lithium salt (LiOTf) into solvents, we report a fluorinated sulfonate electrolyte for application in high-voltage and long-cycle LIBs. The proposed 1.9 M LiFSI/2,2,2-trifluoroethyl trifluoromethanesulfonate-2,2,2-trifluoroethyl electrolyte stabilizes the NCM811 and LCO cathodes under aggressive high-voltage conditions by suppressing parasitic reactions (electrolyte oxidation, transition-metal dissolution, gas evolution, etc.), and enables fast Li+ intercalation/deintercalation kinetics on the graphite anode by forming S-containing species (Li2SOx). Furthermore, the non-flammable electrolyte extends the limits for high-voltage batteries, allowing configurations such as 4.55 V-charged graphite||LCO and 4.6 V-charged graphite||NCM811 to remain stable over thousands of cycles while also maintaining a good safety profile.
Vibrational study of 2,2,2-trifluoroethyl trifluoromethanesulfonate
2,2,2-Trifluoroethyl trifluoromethanesulfonate (CF3SO2OCH2CF3) is a commercially available substance whose molecular structure and vibrational characteristics were not found in the literature. Therefore, it was decided to extend to this substance our previous studies on different derivatives of trifluoromethanesulfonic acid in order to have more information about the conformation characteristics and force constants of this kind of molecules. For that purpose, the infrared and Raman spectra were measured for the substance in different physical states and an optimized molecular structure and frequencies corresponding to the normal modes of vibration were calculated by means of quantum chemistry procedures. 2,2,2-Trifluoroethyl trifluoromethanesulfonate is a commercial product (Oakwood Products, Inc.) and was used with no further purification. It was handled with proper protection from the atmospheric humidity. The infrared spectra were run in a Perkin-Elmer FT–IR instrument, model 1600, and in Bruker FT–IR instruments, models IFS 66 and IFS 113v. The Bruker FRA 106 accessory was used to obtain the Raman spectrum of the liquid substance using light of 1064 nm from Nd/YAG laser for excitation.[2]
In brief, the quantum chemical calculations predict the existence of two stable, asymmetric (point group C1) and structurally equivalent conformers for 2,2,2-Trifluoroethyl trifluoromethanesulfonate, having a gauche conformation. In fact, such a conformation appears as a common feature of YSO2OX sulfonates, including CF3SO2OSiH3 and CF3SO2OSi(CH3)3. Interestingly, in these last two molecules as well as in CF3SO2OCH2CF3, an eclipsed orientation of the OX bond relative to one of the SO double bonds was observed. Infrared and Raman spectra were obtained for 2,2,2-trifluoroethyl trifluoromethanesulfonate, in which bands assignable to 35 out of the expected 39 normal modes of vibration were observed. The optimized molecular geometry, force constants and vibrational frequencies were calculated using DFT techniques in the B3LYP approximation and three different basis set of atomic orbitals. The structural results showed the gauche conformation as the most stable one, in what seems to be a general structural characteristic of covalent sulfonates. Taking as a basis the observed frequencies corresponding to the fundamental vibrations, it was possible to proceed to a scaling of the theoretical force field.
References
[1]Zhang J, Zhang H, Weng S, Li R, Lu D, Deng T, Zhang S, Lv L, Qi J, Xiao X, Fan L, Geng S, Wang F, Chen L, Noked M, Wang X, Fan X. Multifunctional solvent molecule design enables high-voltage Li-ion batteries. Nat Commun. 2023 Apr 18;14(1):2211. doi: 10.1038/s41467-023-37999-4. PMID: 37072401; PMCID: PMC10113204.
[2]Tuttolomondo, M E et al. “Experimental and theoretical vibrational study of 2,2,2-trifluoroethyl trifluoromethanesulfonate, CF3SO2OCH2CF3.” Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy vol. 60,3 (2004): 611-9. doi:10.1016/s1386-1425(03)00269-5
You may like
See also
Lastest Price from 2,2,2-Trifluoroethyl trifluoromethanesulfonate manufacturers
US $2.00-5.00/kg2025-07-17
- CAS:
- 6226-25-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 100kg

US $0.00-0.00/KG2025-07-07
- CAS:
- 6226-25-1
- Min. Order:
- 1KG
- Purity:
- 99.0%
- Supply Ability:
- 10000KGS