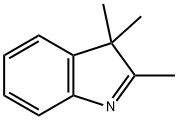
2,3,3-Trimethylindolenine: Building Block for Dyes & Antifungals
2,3,3-Trimethylindolenine is a crucial indole derivative. Its molecular structure is foundational for the synthesis of a wide array of functional molecules. While not typically a pharmacologically active agent itself, its role as a synthetic building block is indispensable. This is particularly true in the creation of cyanine dyes, which are vital reagents for fluorescent labeling and imaging in biological research and clinical diagnostics. Furthermore, derivatives of 2,3,3-trimethylindolenine are explored for their potential in antimicrobial agents and as organic photoconductors, highlighting the versatility of this compound. The primary and most established method for synthesizing 2,3,3-trimethylindolenine is the Fischer indole synthesis. This reaction involves the acid-catalyzed cyclization of a phenylhydrazone, which is formed from the condensation of phenylhydrazine and a ketone. In the case of 2,3,3-trimethylindolenine, the ketone used is 3-methyl-2-butanone (methyl isopropyl ketone).

Microwave Synthesis of Quaternary Ammonium Salts
Fluorescence spectroscopy has become a key technique for the detection and elucidation of biological processes. In particular, cyanine dyes see widespread application as fluorescent probes. The microwave synthesis of several quaternary ammonium salts is described. The synthesis provides comparable or better yields than published methods with reduced reaction times and in the absence of solvent. A simple efficient microwave synthesis of N-alkyl quaternary ammonium salts has now been developed. Reaction times are measured in minutes as opposed to hours and all of the experiments are performed under solvent-free conditions.The reaction of 2,3,3-trimethylindolenine with an array of alkyl halides with varied functionality were studied. The reactions were performed by charging each microwave reaction vial with of 2,3,3-trimethylindolenine and an alkyl halide. Our previously published reaction of ethyl iodide with 2,3,3-trimethylindolenine served as the model system. The microwave reaction conditions were determined using a single-mode microwave system. The temperature was monitored throughout each reaction. The optimized reaction condition was 130 οC, ramp time: 2:50 min, reaction time: 5:00 min giving a 95% yield.[1]
The scope of the reaction was examined with the coupling of 2,3,3-trimethylindolenine and benzothiazole with iodomethane, iodopropane, bromoethanol, and bromohexanoic acid. The hold time, ramp time, and temperature for each electrophile was studied. The optimized reaction conditions are presented. In most cases, the yields were comparable or exceeded the published yields. Most significant is the substantially decreased reaction time and simplicity of the reaction procedure. The yields presented are the yields without resubjection of the filtrates. The single mode microwave system has provided substantially decreased reaction times, simplicity of reaction procedure, and comparable or increased reaction yields observed for reactions conducted.
N-Alkylated 2,3,3-trimethylindolenines in the fight against Saccharomyces cerevisiae infections
The rise in number of immunocompromised patients (either through infections, nutritional irregularities or medical treatment) has led to an increase in opportunistic fungal infections. Inspired by the aforementioned medicinal activity of the benzothiazoles, we have set out to create two small libraries of structurally similar N-alkylated molecules: 2,3,3-trimethylindolenine and 2-methylbenzothiazole. Each of these subclasses has a step-wise increase in linear alkyl chain length (C1–C10). The resulting derivatives show varying degrees of lipopihilicity, as judged by their log P (base 10 logarithm of the partition coefficient) values. The salts of 2,3,3-trimethylindolenine and 2-methylbenzothiazole were prepared by alkylation with the corresponding alkyl/benzyl halides or sultones to afford the compounds in excellent to poor yield. The synthesised compounds were tested in vitro to determine growth inhibitory activity. Minimum inhibitory concentration (MIC) values were determined in sets, by comparison with 2,3,3-trimethylindolenine and 2-methylbenzathiazole (purchased from Alfa Aesar), under the same conditions noted in Tables 1 and
2. It is important to note that the yeast cell surfaces carry a negative charge and thus a good interaction between the yeast cell surface should be observed with the quaternary N-alkylated compounds.[2]
The quaternary N-alkylated derivatives of both 2,3,3-trimethylindolenine and 2-methylbenzothiazole salts show varying degrees of antifungal activity with the longer chain substituents being more potent against S. cerevisiae and S. pombe. We propose that the growth inhibition can be attributed to the charged N-alkylated compounds with the increasing linear chain lengths. We postulate that these compounds are attracted to the negatively charged yeast membrane, with the longer lipophilic chains being absorbed into and subsequently distorting the lipid bilayer. Compounds all show MIC values of <35 μg/mL and are thus deemed from this study to be the most potent towards S. cerevisiae and S. pombe. Together with further compounds, which are being generated with higher degrees of lipophilicity these compounds will be taken forward for mammalian screening. This response may be the result of C. albicans' ability to adapt to the antifungal stress and develop drug tolerance. As a diploid fungus, C. albicans contains a large number of drug exclusion mechanisms with varying substrate specificities enabling it to obtain tolerance to many novel antifungals, a resistance not seen with the haploid yeast species, S. cerevisiae and S. pombe. The results highlighted above imply that longer N-alkylated (i.e. >C7) 2,3,3-trimethylindolenines and 2-methylbenzothiazole salts show potential for selective targeting towards S. cerevisiae infections which are a major problem in health care.
Synthesis of Near-Infrared Fluorescent Indole-Based Squaraines
Since the first reports on the use of microwave (MW) heating to accelerate organic chemical transformations by the groups of Gedye and Giguere and Majetich in 1986, microwave-assisted organic synthesis (MAOS) has proven to be a powerful technique for promoting a variety of chemical reactions. Herein, we report a common synthetic pathway for the preparation of a wide color range of symmetrical and unsymmetrical 2,3,3-trimethylindolenine-based squaraine dyes, using MW methodologies, which offers a practical approach to the rapid preparation of a variety of squaraines. Reaction time under MW was reduced from days to minutes, with more than 2-fold improvement in product yields when compared to conventional methods. Crystallization methods were developed on the crude symmetrical and unsymmetrical products, while some unsymmetrical dyes were isolated with good purity only after column chromatography. A microwave-assisted method for the preparation of a wide color range of 2,3,3-trimethylindolenine-based squaraines and their intermediates is described. This practical approach allows the rapid preparation of both symmetrical and nonsymmetrical squaraine dyes, reducing reaction time from days to minutes with more than 2-fold improvement in product yields when compared to conventional methods.[3]
References
[1]Winstead AJ, Fleming N, Hart K, Toney D. Microwave synthesis of quaternary ammonium salts. Molecules. 2008 Sep 3;13(9):2107-13. doi: 10.3390/molecules13092107. PMID: 18830143; PMCID: PMC6245181.
[2]Tyler, A. R., Okoh, A. O., Lawrence, C. L., Jones, V. C., Moffatt, C., & Smith, R. B. (2013). N-alkylated 2,3,3-trimethylindolenines and 2-methylbenzothiazoles. Potential lead compounds in the fight against Saccharomyces cerevisiae infections. European Journal of Medicinal Chemistry, 64, 222–227.
[3]Barbero, Nadia et al. “Microwave-Assisted Synthesis of Near-Infrared Fluorescent Indole-Based Squaraines.” Organic letters vol. 17,13 (2015): 3306-9. doi:10.1021/acs.orglett.5b01453
You may like
Related articles And Qustion
Lastest Price from 2,3,3-Trimethylindolenine manufacturers
US $0.00/kg2025-10-28
- CAS:
- 1640-39-7
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $10.00/KG2025-04-21
- CAS:
- 1640-39-7
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt