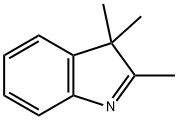
2,3,3-Trimethylindolenine: Synthesis & Applications
2,3,3-Trimethylindolenine is a compound used as a reactant in organic synthesis reactions. At ambient temperatures, it exists as a white to pale yellow crystalline powder possessing a strong odour reminiscent of jasmine, orange blossom, or civet. As a vital raw material for perfumes and flavourings, it finds extensive application in the perfume, cosmetics, and food industries, imparting a long-lasting floral note to products. 2,3,3-Trimethylindolenine can be synthesised via the Fischer indole synthesis method, exhibiting excellent stability and fixative properties, making it an indispensable key component in the fragrance industry.

Divergent Synthesis of 2,3,3-Trimethylindolenine and Indoline Ring Systems
2,3,3-Trimethylindolenines bearing a C3-quaternary stereocenter and indolines bearing a C2-quaternary stereocenter are widely occurring core structures in many natural products and biologically active molecules, and the synthetic methods that enable efficient construction of these core structures is in high demanded. Catalytic asymmetric dearomatization (CADA) reactions serve as one of the most straightforward approaches to chiral building blocks from aromatic substrates. Recently, a series of CADA reactions have been developed for the construction of chiral 2,3,3-Trimethylindolenine/indoline derivatives based on the reactivity of C(sp2)–Pd species. The majority of these reactions were performed in an intramolecular manner, in which a haloarene or an acetylene is tethered to the N1-, C2- or C3-position of indole as the C(sp2)–Pd precursor. The You group achieved enantioselective C3-arylation of C3-tethered indoles; Jia and Fukuyama developed the dearomative Heck reaction of N1- and C2-tethered indoles to build a C2-stereocenter; Jia, Liang, Lautens, and Dai reported the asymmetric dearomative difunctionalization of N1-tethered indoles by employing an external nucleophile.[1]
Compared with intramolecular cyclization, the intermolecular version allows for a more divergent approach to dearomatized products. In this regard, Zhang and co-workers reported recently an enantioselective annulative dearomatization of C3-bromoarylindoles with alkynes using the Pd/Sadphos catalytic system to access various spiro-2,3,3-Trimethylindolenines with a C3-quaternary stereocenter. However, to date the intermolecular asymmetric dearomatization of C2-arylindoles has not been reported yet, and stereoselective construction of structurally diversified indoline/2,3,3-Trimethylindolenine derivatives remains a formidable challenge. Herein, we report a Pd-catalyzed asymmetric intermolecular spiroannulation reaction of C2-arylindoles with internal alkynes, leading to the formation of spiro-indolines bearing a C2-quaternary stereocenter. These products could undergo a stereospecific aza-semipinacol rearrangement under acidic conditions, affording fused-2,3,3-Trimethylindolenines with a C3-quaternary stereocenter. Furthermore, the migration selectivity of this process could be tuned by the selection of reaction sequence. The asymmetric spiroannulation together with the stereospecific rearrangement enabled a divergent access to dearomatized indole derivatives with either a C3- or a C2-quaternary stereocenter. In summary, we have developed a palladium-catalyzed enantioselective intermolecular dearomatization of C2-arylindoles with internal alkynes, leading to C2-spiroindolines bearing a C2-quaternary stereocenter with good yields and enantioselectivities. The stereospecific aza-semipinacol rearrangement afforded enantioenriched 2,3,3-Trimethylindolenine derivatives bearing a C3-quaternary stereocenter via a tunable aryl/alkenyl migration. The combined steps enabled a divergent access to dearomatized indole derivatives with either a C3- or a C2-quaternary stereocenter.
Synthesis of indole derivatives as prevalent moieties
Indoles are a significant type of heterocycle as they are found in proteins in the form of amino acids, such as tryptophan. They are also present in several drugs, such as indomethacin and the notorious LSD, and several plants such as strychnine. The incorporation of an indole core, a biologically known pharmacophore in medicinal molecules, means it is a useful heterocyclic that can bear a number of biological properties. Compounds containing the indole nucleus exhibit several different biological properties, including anti-cancer, anti-fungal, anti-HIV, anti-inflammatory, anti-viral, anti-tubercular, anti-microbial, anti-hypertensive, and anti-diabetic activities, and also photochemotherapeutic properties. In addition, under acidic reaction conditions, upon the protonation of spiroindoleninpyrroloquinazolinone 23 on the 2,3,3-Trimethylindolenine nitrogen, underwent a retrograde aldol reaction to afford the ring-opened pyrroloquinazolinone. In 2010, Danishefsky et al. demonstrated the Pictet–Spengler reaction for C2-aryl indoles, and effectively separated the elusive azaspiro 2,3,3-Trimethylindolenine intermediate of the Pictet–Spengler reaction. Total synthesis of phalarine (63) was commenced from β-carboline 56 and after 11 steps afforded the intermediate 57. The latter, using p-toluenesulfonic acid in toluene, gave the corresponding indole product 58 (5% yield), benzofuro[3,2-b]indole 59 (17% yield), benzofuro[3,2-b]indol-8-amine 60 (18% yield), and diindole-2-carboxylate 61 (9% yield). In summary, indoles represent one of the most significant privileged motifs in drug discovery. Indoles and their derivatives have the exclusive property of mimicking the structure of peptides and can bind reversibly to enzymes, giving incredible opportunities to identify unique drugs that possess various modes of action.[2]
References
[1]Gao, Dong, and Lei Jiao. “Divergent Synthesis of Indolenine and Indoline Ring Systems by Palladium-Catalyzed Asymmetric Dearomatization of Indoles.” Angewandte Chemie (International ed. in English) vol. 61,13 (2022): e202116024. doi:10.1002/anie.202116024
[2]Heravi MM, Amiri Z, Kafshdarzadeh K, Zadsirjan V. Synthesis of indole derivatives as prevalent moieties present in selected alkaloids. RSC Adv. 2021 Oct 15;11(53):33540-33612. doi: 10.1039/d1ra05972f. PMID: 35497516; PMCID: PMC9042329.
You may like
Related articles And Qustion
See also
Lastest Price from 2,3,3-Trimethylindolenine manufacturers
US $0.00/kg2025-10-28
- CAS:
- 1640-39-7
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $10.00/KG2025-04-21
- CAS:
- 1640-39-7
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt