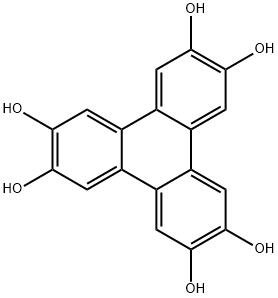
2,3,6,7,10,11-Triphenylenehexol: Building Block for Conductive MOFs & COFs
2,3,6,7,10,11-Triphenylenehexol is a polyhydroxy aromatic compound featuring a triphenylene fused-aromatic core with six hydroxyl groups symmetrically substituted, exhibiting good thermal stability and an electron-rich aromatic structure. Its molecule adopts a planar disc-like conformation, with the six hydroxyl groups providing abundant reactive sites. It can self-assemble via covalent or coordinate bonds, making it a key monomer for constructing two-dimensional covalent organic frameworks (COFs) and metal-organic frameworks (MOFs). 2,3,6,7,10,11-Triphenylenehexol is also used in the preparation of liquid crystal materials, fluorescent probes and organic optoelectronic functional materials, and finds widespread application in cutting-edge materials fields such as energy storage, catalysis and electronic devices.

An Electrically Conducting Three‐Dimensional Iron–Catecholate Porous Framework
In recent years, electrical conductivity has been added to the property portfolio of metal–organic framework (MOF) materials. This exciting new feature in combination with the central properties of MOFs, including high crystallinity, porosity and enormous diversity of molecular building blocks, is expected to pave the way for new directions in fields such as charge storage, sensing and photovoltaic materials. The most prominent of these materials is the metal catecholate, M‐CAT‐1 series consisting of 2,3,6,7,10,11-Triphenylenehexol (HHTP) moieties and metal ions, such as CoII, NiII or CuII. The structure of Fe‐CAT‐5 is built from iron and HHTP bridging units that form a 2‐fold interpenetrating network with srs topology in which sulfate and dimethylammonium ions are part of the framework. Although these frameworks conduct protons well at higher relative humidity, their electrical conductivity remains unknown. Recently, Dinca and co‐workers reported the synthesis of cubic faujasite‐like, rare‐earth‐metal 2,3,6,7,10,11-Triphenylenehexol MOFs with intrinsic electrical conductivity values of up to 10−5 S cm−1. Nevertheless, to date, the combination of the transition metal iron with the well‐known 2,3,6,7,10,11-Triphenylenehexol building unit to give a non‐interpenetrating 3D catecholate‐based framework with a defined structure, high porosity and high conductivity remains elusive. We now present the synthesis of a novel metal–organic framework, coined Fe‐HHTP‐MOF, comprising the trigonal planar building block 2,3,6,7,10,11‐hexahydroxytriphenylene and FeIII ions.[1]
Herein, we present the synthesis of a cubic ferric catecholate framework material coined Fe‐2,3,6,7,10,11-Triphenylenehexol‐MOF. The Fe‐HHTP‐MOF is obtained in a solvothermal reaction as a dark black powder consisting of intergrown tetrahedral crystallites in the 300–500 nm size range. The structure was solved from powder X‐ray diffraction data using a Mo‐K α source by indexing and a subsequent simulated annealing step. The unit cell obtained was subjected to a DFT optimization and Rietveld refinement to give a final structure model with the cubic space group F23. The structure of Fe‐2,3,6,7,10,11-Triphenylenehexol‐MOF is composed of iron‐connected supertetrahedra to give a diamond‐like topology. The tetrahedra are composed of four triphenylene units that form the facets and are interconnected by a defined trinuclear iron–oxo cluster. The Fe‐HHTP‐MOF is microporous with a BET surface area exceeding 1400 m2 g−1. Moreover, Fe‐HHTP‐MOF combines this high surface area with electrical conductivity of about 10−3 S cm−1, thus establishing an exceptionally high intrinsic conductivity for a 3D‐connected framework. The combination of the substantial porosity of Fe‐2,3,6,7,10,11-Triphenylenehexol‐MOF with high electrical conductivity and a narrow direct band gap offers exciting opportunities for the design of novel chemical sensors, organic optoelectronics, and for electric control of mass transport in porous systems, such as microfluidics.
2,3,6,7,10,11-Triphenylenehexol for the synthesis of group 13 MOFs
In the search of 3D Al-MOFs containing polyphenolate linker molecules we have considered the use of the polycatecholate 2,3,6,7,10,11-Triphenylenehexol employing our high-throughput synthesis techniques. This linker is very well known for its redox activity – it can adopt seven different redox states – and has been employed in the synthesis of complexes, covalent-organic frameworks (COFs) and very few MOFs like CAT-1 and CAT-5. The layered material CAT-1 has been described with divalent Ni2+ and Co2+ ions and contains radical anions as well as fully deprotonated HHTP6− ions. In the structures of Fe(iii)-, Ti(iv)- and V(iv)-CAT-5 the octahedrally coordinated isolated metal ions are each connected through three HHTP6− ions. Charge balance is accomplished through incorporation of DMA+ ions into the pores which results in structures with limited porosity. Herein we report the synthesis of ((CH3)2NH2)2[Al3O(HHTP)(HHTP˙)]·0.5DMF·18H2O and ((CH3)2NH2)2[Ga3O(HHTP)(HHTP˙)]·14.5H2O, using 2,3,6,7,10,11-Triphenylenehexol (H6HHTP) as linker. The structure was determined by a combination of continuous rotation electron diffraction (cRED), structure modelling and Density Functional Theory (DFT) calculations. The resulting compounds were thoroughly characterized by thermogravimetric analyses (TGA), elemental analyses (EA), variable temperature powder X-ray diffraction (VT-PXRD) as well as infrared (IR), 1H and 27Al solid-state nuclear magnetic resonance (NMR) and electron paramagnetic resonance (EPR) spectroscopy.[2]
In conclusion we synthesized two new isostructural MOFs (Al3+ and Ga3+ based) with a β-cristobalite network type. The utilization of 2,3,6,7,10,11-Triphenylenehexol as a linker under the given synthetic conditions led to implementation of six-fold deprotonated (HTTP6−) and partly radical ligands (HHTP˙3−). The two-fold negative network charge is most likely compensated by dimethylammonium ions which are located close to the trimeric IBU. Due to the radical character of half of the linker molecules potential applications in redox chemistry might be possible and should be the aim of further investigations.
Efficient electroorganic synthesis of 2,3,6,7,10,11-Triphenylenehexol derivatives
The unique spectroscopic and geometric features of triphenylenes give rise to a variety of applications for this very common structural motif. The use of triphenylenes in discotic liquid crystals as building blocks in supramolecular chemistry as well as in solid-state chemistry is well documented. Since we have a specific interest in a large-scale access to derivatives of 2,3,6,7,10,11-Triphenylenehexol, we were prompted to develop a sustainable protocol providing the target compound by electroorganic methods. Triphenylene ketals are easily available by anodic oxidation using a simple galvanostatic protocol. Best results are obtained when the synthesized dehydrotrimers are almost insoluble in the electrolyte. Precipitation occurs during the electrolysis when employing tetraalkylammonium salts in PC. This effectively prevents the desired products from over-oxidation. Since PC is environmentally benign and inexpensive, the procedure significantly improved the access to the triphenylene derivatives. Moreover, acid-catalyzed cleavage of triphenylene ketals provides 2,3,6,7,10,11-Triphenylenehexol almost quantitatively in very good quality.[3]
References
[1]Mähringer A, Döblinger M, Hennemann M, Gruber C, Fehn D, Scheurle PI, Hosseini P, Santourian I, Schirmacher A, Rotter JM, Wittstock G, Meyer K, Clark T, Bein T, Medina DD. An Electrically Conducting Three-Dimensional Iron-Catecholate Porous Framework. Angew Chem Int Ed Engl. 2021 Aug 9;60(33):18065-18072. doi: 10.1002/anie.202102670. Epub 2021 Jul 1. PMID: 33780115; PMCID: PMC8456825.
[2]Leubner, S et al. “Hexahydroxytriphenylene for the synthesis of group 13 MOFs - a new inorganic building unit in a β-cristobalite type structure.” Dalton transactions (Cambridge, England : 2003) vol. 49,10 (2020): 3088-3092. doi:10.1039/d0dt00235f
[3]Regenbrecht C, Waldvogel SR. Efficient electroorganic synthesis of 2,3,6,7,10,11-hexahydroxytriphenylene derivatives. Beilstein J Org Chem. 2012;8:1721-4. doi: 10.3762/bjoc.8.196. Epub 2012 Oct 10. PMID: 23209505; PMCID: PMC3511005.
See also
Lastest Price from 2,3,6,7,10,11-Triphenylenehexol manufacturers
US $0.00-0.00/KG2025-04-04
- CAS:
- 4877-80-9
- Min. Order:
- 1KG
- Purity:
- 98%
- Supply Ability:
- 1ton
US $1.00-2.10/KG2024-08-08
- CAS:
- 4877-80-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 20000KG