2,6-Dioxopiperidine-3-ammonium Chloride: Synthesis & Pharmaceutical Applications
2,6-Dioxopiperidine-3-ammonium chloride readily dissolves in DMSO and methanol, with limited solubility in water. It requires storage under inert gas atmosphere, sealed in a dry, light-protected container at room temperature to prevent moisture absorption and decomposition. Its core application lies as a critical synthetic intermediate for immunomodulatory and antitumour drugs such as lenalidomide and pomalidomide. Utilising this compound as a starting material, the phthalimide skeleton can be constructed through condensation and reduction steps, enabling efficient synthesis of drug molecules. 2,6-Dioxopiperidine-3-ammonium chloride is also employed in preparing target protein degradation conjugates, supporting novel drug development pathways such as PROTAC. As a critical building block in pharmaceutical synthesis, its high-purity form directly impacts drug yield and quality. It finds extensive application in anti-tumour drug development, active pharmaceutical ingredient production, and chemical biology research.

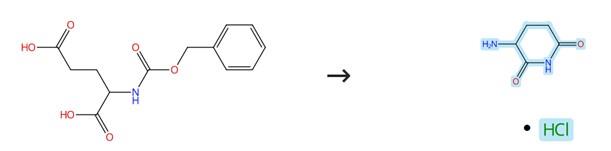
Synthesis of 2,6-Dioxopiperidine-3-ammonium chloride from Benzyl (2,6-dioxopiperidin-3-yl)carbamate
2,6-Dioxopiperidine-3-ammonium chloride can be prepared as the following steps: benzyloxy carbonyl1-L-glutamine A solution of sodium hydroxide (27.4 g) in water (300 mL) was added at 25°C to 30°C to a mixture of L-glutamine (100 g) and toluene (300 mL) to obtain a reaction mixture. Benzyl chloroformate (50% solution in toluene; 420 g) was added to the reaction mixture slowly over a period of 60 minutes to 90 minutes while maintaining the pH to 11.0 to 12.0 with simultaneous addition of aqueous sodium hydroxide solution (120 mL; 31.5 g sodium hydroxide dissolved in 120 mL of de-ionized water). Water was added to the reaction mixture and the layers were separated. The aqueous layer was washed with toluene (300 mL). The pH of the aqueous layer was adjusted to 1.0 to 1.5 with dilute hydrochloric acid (100 mL) to obtain a slurry. The slurry was cooled to 0°C to 5°C and then stirred for one hour. The product so obtained was filtered, washed with water (2x200 mL) to obtain a wet solid. The wet solid was dried in an air oven at 50°C to 55°C to obtain 2,6-Dioxopiperidine-3-ammonium chloride.[1]
Synthesis of 2,6-Dioxopiperidine-3-ammonium chloride using 3-Amino-piperidine-2,6-dione
In a 10 L, 3 necked RBF N-benzyloxycarbonyl-3-amino-3-methylpiperidine-2, 6-dione, (175.40 g) was suspended in methanol (203 1 .69 mL). To this suspension was added 4 N HCI (173.88 mL), followed by 10% Pd / C catalyst (5g, 0.0075 eq). The mixture was hydrogenated under 1 atm of hydrogen (balloon) for 23 hours. Hydrogenation was stopped and the suspension was vacuum filtered through a weighed Buchner funnel. The clear filtrate was fed to a pre-weighed flask on a rotary evaporator and evaporated to an aqueous crystal slurry. The solid from the filter funnel was suspended in hot water. After evaporation Isopropanol ( 150 mL) was added to the aqueous slurry and the slurry was filtered. After drying (4h at 60°C) 89.02 g (80.9% ) of the title compound was obtained. The aqueous crystal suspension from the evaporated methanol filtrates were combined with the aqueous isopropanol filtrate from solid workup, and to this additional dry Isopropanol ( 100.00 mL) was added. The slurry was concentrated again on the rotavapor and filtered. The wet solid (28.31 g) was dried in vacuum for 74 h to a mass of 16.61 g. A total mass of 105.61 (96% ) of pure 2,6-Dioxopiperidine-3-ammonium chloride was obtained.[2]
2,6-Dioxopiperidine-3-ammonium chloride applied in chemical synthesis
2,6-Dioxopiperidine-3-ammonium chloride can serve as a key intermediate for lenalidomide and its related derivatives. Lenalidomide, an anticancer drug, was synthesized using greener reaction conditions. Unlike conventional methods that utilize egregious organic solvents and expensive reagents or catalysts, our approach employs ecofriendly reaction media, cost-effective reagents, and simplified downstream processing. These modifications lead to improved green chemistry metrics, as reflected by a favorable complete E-factor (cEF) and process mass intensity (PMI). The optimized process has been successfully scaled up, affording an overall isolated yield of 62%. One step involved the cyclocondensation of intermediate with 2,6-Dioxopiperidine-3-ammonium chloride. The reaction was performed under aqueous micellar conditions; however, along with the desired product, a side reaction producing 4-nitrophthalide was also observed. Methyl 2-(bromomethyl)-3-nitrobenzoate (15.0 g, 54.9 mmol), 2,6-Dioxopiperidine-3-ammonium chloride (9.0 g, 54.9 mmol), and N,N-diisopropylethylamine (38.2 mL, 219.6 mmol) were combined in a 100 mL glass vessel equipped with a PTFE-coated magnetic stir bar. The mixture was stirred at 80–85 °C for 12 h. After completion (monitored by TLC), the reaction mixture was cooled to room temperature and filtered through a sintered funnel, and the solid was washed with cold ethanol (25 mL). The filter cake was dried under reduced pressure to give 13.6 g of compound as a gray solid (86% yield, uncorrected), purity 95.73% (by HPLC).[3]
References
[1]WO2018154516A1
[2]WO2011050962
[3]Wandre, R. S., Sharma, K., Pathan, A. M., Nabi, M., & Gade, A. B. (2025). Environmentally benign synthesis of anticancer drug lenalidomide. Organic Process Research & Development, 29(11), 2984–2988.
You may like
Related articles And Qustion
Lastest Price from 2,6-Dioxopiperidine-3-ammonium chloride manufacturers

US $0.00/kg2026-03-03
- CAS:
- 24666-56-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 99999

US $0.00-0.00/kg2025-08-29
- CAS:
- 24666-56-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1